
Chemistry, 7e (McMurry/Fay)
Chapter 11 Liquids, Solids, and Phase Changes
11.1 Multiple-Choice Questions
1) When a narrow diameter glass tube is inserted into a body of water, water rises in the tube and its
surface inside is concave upwards. Which statement, concerning the strength of the intermolecular forces
between glass and water molecules compared to those between water molecules, is accurate?
A) The forces of attraction between the glass and water are weaker than those in water.
B) The forces of attraction between the glass and water are stronger than those in water.
C) The forces of attraction between the glass and water are the same as those in water.
D) Intermolecular forces are irrelevant to this situation.
2) Which of the intermolecular forces is the most important contributor to the high surface tension shown
by water?
A) dipole-dipole forces
B) dispersion forces
C) hydrogen bonding
D) ion-dipole forces
3) Which of the following is expected to have the greatest viscosity?
A) C5H12
B) C6H14
C) C5H11OH
D) CH4

4) The magnitude of the heats of vaporization, fusion and sublimation of a substance reflect the
A) density of the substance.
B) magnitudes of the boiling and melting points of the substance.
C) strength of the covalent bonds between atoms in each molecule of the substance.
D) strength of the intermolecular forces of the substance.
5) For a particular compound, which is expected to be the largest in general?
A) the heat required to raise the temperature of one mole of the gas 10.0°C
B) the heat required to raise the temperature of one mole of the liquid 10.0°C
C) the molar heat of fusion at the normal melting point
D) the molar heat of vaporization at the normal boiling point
6) When a substance melts at its normal melting point, the sign of ΔH is ________ and the sign of ΔS of
this phase change is ________.
A) +, -
B) -, +
C) +, +
D) -, -
7) Ethyl chloride, C2H5Cl, is used as a local anesthetic. It works by cooling tissue as it vaporizes. The heat
of vaporization is 26.4 kJ/mol. How much heat could be removed by 60.0 g of ethyl chloride?
A) 24.6 kJ
B) 1584 kJ
C) 2.28 kJ
D) 1703 kJ

8) Bromine is one of only two elements that is a liquid at room temperature. Bromine has a heat of
vaporization of 30.91 kJ/mol and its boiling point is 59°C. What is the entropy of vaporization for
bromine?
A) -301 J/(mol ∙ K)
B) -93.1 J/(mol ∙ K)
C) 10.7 J/(mol ∙ K)
D) 93.1 J/(mol ∙ K)
9) CFC-11 (trichlorofluoromethane, CCl3F) has been used for many years as the working fluid in
refrigerators. Given its heat of vaporization is 26.88 kJ/mol and its entropy of vaporization is 90.51 J/(mol ∙
K), what is the boiling point of CFC-11?
A) -272.9°C
B) 0.297°C
C) 2.44°C
D) 23.8°C
10) How much heat is released when 125.0 g of steam at 100.0°C is cooled to ice at -15.0°C? The enthalpy
of vaporization of water is 40.67 kJ/mol, the enthalpy of fusion for water is 6.01 kJ/mol, the molar heat
capacity of liquid water is 75.4 J/(mol ∙ °C), and the molar heat capacity of ice is 36.4 J/(mol ∙ °C).
A) 91.3 kJ
B) 325 kJ
C) 380 kJ
D) 413 kJ
11) For the process: HNO3(g) ⇌ HNO3(l), ΔH° is -39.04 kJ/mol and ΔS° is -111.74 J/(mol ∙ K). What is the
normal boiling point of pure HNO3?
A) 2.86°C
B) 76.2°C
C) 270.3°C
D) 349.4°C

12) Which of the following best explains why ΔHvap is usually higher than ΔHfusion?
A) Vaporization occurs at high temperature.
B) Vaporization involves the breaking of bonds within molecules.
C) Vaporization involves the breaking of all bonds between molecules.
D) Vaporization increases the entropy of molecules.
13) As a liquid evaporates at a temperature below its boiling point, the temperature of the liquid
A) decreases.
B) decreases at low temperatures, but increases at high temperatures.
C) increases.
D) remains unchanged.
14) Molecules of a liquid can pass into the vapor phase only if the
A) liquid has little surface tension.
B) molecules have sufficient kinetic energy to overcome the intermolecular forces in the liquid.
C) temperature of the liquid is near its boiling point.
D) vapor pressure of the liquid is high.
15) The vapor pressure of a pure liquid increases as the
A) average kinetic energy of the molecules in the liquid phase decreases.
B) intermolecular attractive forces increase.
C) temperature of the liquid phase decreases.
D) temperature of the liquid phase increases.

16) The normal boiling point occurs when the
A) intermolecular forces within the liquid phase are broken.
B) temperature of the pure liquid equals the external temperature.
C) vapor pressure of a pure liquid equals an external pressure of one atmosphere.
D) vapor pressure of the liquid equals the external pressure.
17) Which of the following compounds has the highest boiling point?
A) CH3CH2OH
B) HOCH2CH2OH
C) H3C—O—CH3
D) CH3CH2CH2CH3
18) The normal boiling point for HBr is higher than the normal boiling point for HCl. This can be
explained by
A) larger dipole-dipole forces for HBr.
B) larger dispersion forces for HBr.
C) larger hydrogen-bond forces for HBr.
D) larger dipole-dipole forces, larger dispersion forces, and larger hydrogen-bond forces for HBr.
19) While mercury is very useful in barometers, mercury vapor is toxic. Given that mercury has a ΔHvap
of 59.11 kJ/mol and its normal boiling point is 356.7°C, calculate the vapor pressure in mm Hg at room
temperature, 25°C.
A) 2.68 × 10-3 mm Hg
B) 2.99 mm Hg
C) 372 mm Hg
D) 753 mm Hg

20) Hydroquinone is an antioxidant that is also used as a photographic reducer and developer. The
normal boiling point of hydroquinone is 310°C. Calculate the pressure at which hydroquinone will boil at
200°C given that its ΔHvap is 73.38 kJ/mol.
A) 1.210 × 10-4 mm Hg
B) 1.35 mm Hg
C) 22.5 mm Hg
D) 757 mm Hg
21) The vapor pressure of liquid chloroform, CHCl3, is 400.0 torr at 24.1°C and 100.0 torr at -6.3°C. What
is ΔHvap of chloroform?
A) 15.3 kJ/mol
B) 30.1 kJ/mol
C) 57.6 kJ/mol
D) 86.7 kJ/mol
22) Which of the following liquids will exhibit the highest vapor pressure?
A) Br2, bp = 58.8°C
B) CH3OH, bp = 64.7°C
C) H2O, bp = 100°C
D) All exhibit the same vapor pressure.
23) Which of the following statements is not consistent with the properties of a molecular solid?
A) a compound that conducts electricity when molten
B) a low melting solid
C) a solid formed by the combination of two nonmetallic elements
D) a solid that is a nonconductor of electricity

24) A crystalline solid of unknown origin forms an aqueous solution that conducts an electrical current.
The solid has a high melting point and shatters when struck with a hammer. The solid is likely to be
A) a covalent network solid.
B) an ionic solid.
C) a metallic solid.
D) a molecular solid.
25) The wavelength of light used to observe an object must be ________ than the object itself.
A) larger
B) smaller
C) of higher energy
D) of lower energy
26) The structure of a solid can be determined by diffraction of radiation in which region of the
electromagnetic radiation spectrum?
A) infrared
B) microwave
C) visible
D) X-ray
27) How many atoms are in one face-centered cubic unit cell of a metal?
A) 1
B) 2
C) 3
D) 4

28) How many atoms are in one body-centered cubic unit cell of a metal?
A) 1
B) 2
C) 3
D) 4
29) When cubic unit cells stack together, how many unit cells share a common corner?
A) 2
B) 4
C) 6
D) 8
30) How many unit cells share an atom that is on the face of a face-centered cubic unit cell?
A) 1
B) 2
C) 4
D) 8
31) Which type of spherical packing has the most unused space?
A) body-centered cubic
B) cubic closest-packed
C) cubic closest-packed and hexagonal closest-packed
D) simple cubic

32) Iron crystallizes in a body-centered cubic cell having an edge length of 287 pm. What is the density of
iron in g/cm3?
A) 1.99
B) 7.85
C) 11.9
D) 15.9
33) What is the edge length of a face-centered cubic unit cell made up of atoms having a radius of 200
pm?
A) 71 pm
B) 566 pm
C) 20 pm
D) 110 pm
34) Palladium has a face-centered cubic structure and has a density of 12.023 g/cm3. What is its atomic
radius?
A) 388 pm
B) 151 pm
C) 220 pm
D) 245 pm

35) Iridium crystallizes in a face-centered cubic structure. What is the edge length of the unit cell if the
atomic radius of silver is 180 pm?
A) 441 pm
B) 64 pm
C) 38 pm
D) 509 pm
36) An element forms a body-centered cubic crystalline substance. The edge length of the unit cell is 287
pm and the density of the crystal is 7.92 g/cm3. Calculate the atomic weight of the substance.
A) 45.0 amu
B) 48.0 amu
C) 56.4 amu
D) 63.5 amu
37) Lead has a radius of 154 pm and crystallizes in a face-centered cubic unit cell. What is the edge length
of the unit cell?
A) 35 pm
B) 54 pm
C) 1232 pm
D) 436 pm
38) Chromium crystallizes in a body-centered cubic structure. What is the coordination number of each
atom?
A) 4
B) 6
C) 8
D) 12

39) Cubic closest-packing
A) has a body-centered cubic unit cell.
B) has a face-centered cubic unit cell.
C) has a simple cubic unit cell.
D) has the same unit cell as hexagonal closest-packing.
40) The highest coordination number for spherical packing is found in the
A) body-centered cubic structure.
B) simple cubic structure.
C) body-centered cubic and face-centered cubic.
D) cubic closest-packing and hexagonal closest packing.
41) KBr crystallizes in a cubic unit cell with Br- ions on each corner and each face. How many K+ ions and
Br- ions are in each unit cell of KBr?
A) 1 K+ ion and 1 Br- ion
B) 2 K+ ions and 2 Br- ions
C) 4 K+ ions and 4 Br- ions
D) 8 K+ ions and 8 Br- ions
42) An ionic compound crystallizes in a unit cell having a face-centered cubic array of anions, X-, and half
of the tetrahedral holes filled with metal ions, Mn+ The empirical formula of this ionic compound is
A) MX.
B) MX2.
C) M2X.
D) M2X7.

43) An ionic compound crystallizes in a unit cell having a face-centered cubic array of metal ions, Mn+,
and all of the tetrahedral holes occupied by anions, X-. The empirical formula of this ionic compound is
A) MX.
B) MX2.
C) M2X.
D) M7X4.
44) The edge length of a face-centered cubic lattice of NaCl is 564 pm. What is the density of NaCl in
g/cm3?
A) 0.720
B) 1.08
C) 2.16
D) 4.32
45) How many Br- ions are around each K+ ion in KBr, which has a cubic unit cell with Br- ions on each
corner and each face?
A) 1
B) 4
C) 6
D) 8

46) A binary ionic compound, MxAy, crystallizes in a cubic structure that contains eight anions (A)
entirely within its unit cell and a cation (M) on each corner and on each face. What is the empirical
formula of this compound?
A) MA
B) MA2
C) M2A
D) M4A8
47) O2 and O3 are ________ of oxygen.
A) allotropes
B) isomers
C) isotopes
D) stereomers
48) The critical temperature of a substance is the
A) highest temperature at which the liquid phase can exist in equilibrium with the gas phase.
B) temperature above which the compound decomposes.
C) temperature at which all three phases can exist in equilibrium.
D) temperature at which sublimation occurs.
49) Which transition could occur if a solid is heated at a pressure below the triple point pressure?
A) condensation
B) deposition
C) melting
D) sublimation

50) A supercritical fluid refers to a substance
A) above both its critical temperature and its critical pressure.
B) at its triple point.
C) that is in the liquid crystal state.
D) with a viscosity of zero.
51) A safe method of removing caffeine from coffee is through the use of
A) supercritical carbon dioxide.
B) benzene.
C) methylene chloride.
D) water.
52) If figure (1) represents the vapor pressure of water at 25°C, which figure represents the vapor
pressure of water at 45°C?
A) figure (2)
B) figure (3)
C) figure (4)

53) If figure (1) represents the vapor pressure of water at 25°C in a 1 liter container, which figure
represents the vapor pressure of water at 25°C in a 2 liter container?
A) figure (2)
B) figure (3)
C) figure (4)
54) If figure (1) represents the vapor pressure of water at 25°C, which figure represents the vapor
pressure of ethanol, CH3CH2OH at 25°C?
A) figure (2)
B) figure (3)
C) figure (4)

55) If figure (1) represents the vapor pressure of diethyl ether, CH3CH2OCH2CH3, at 25°C, which figure
represents the vapor pressure of ethanol at 25°C?
A) figure (2)
B) figure (3)
C) figure (4)
56) If figure (1) represents the vapor pressure of water, at 25°C, which figure represents the vapor
pressure of mercury at 25°C?
A) figure (2)
B) figure (3)
C) figure (4)
57) Based on the relative strengths of the intermolecular forces of attraction of each substance, which is
the most likely vapor pressure vs. temperature curve for diethyl ether?
A) curve (a)
B) curve (b)
C) curve (c)
D) curve (d)

58) Based on the relative strengths of the intermolecular forces of attraction of each substance, which is
the most likely vapor pressure vs. temperature curve for ethanol?
A) curve (a)
B) curve (b)
C) curve (c)
D) curve (d)
59) Based on the relative strengths of the intermolecular forces of attraction of each substance, which is
the most likely vapor pressure vs. temperature curve for mercury?
A) curve (a)
B) curve (b)
C) curve (c)
D) curve (d)
60) Based on the relative strengths of the intermolecular forces of attraction of each substance, which is
the most likely vapor pressure vs. temperature curve for water?
A) curve (a)
B) curve (b)
C) curve (c)
D) curve (d)

61) From the plot of vapor pressure as a function of temperature shown below, the normal boiling point
for tert-butyl alcohol is approximately
A) 0°C.
B) 40°C.
C) 85°C.
D) 100°C.
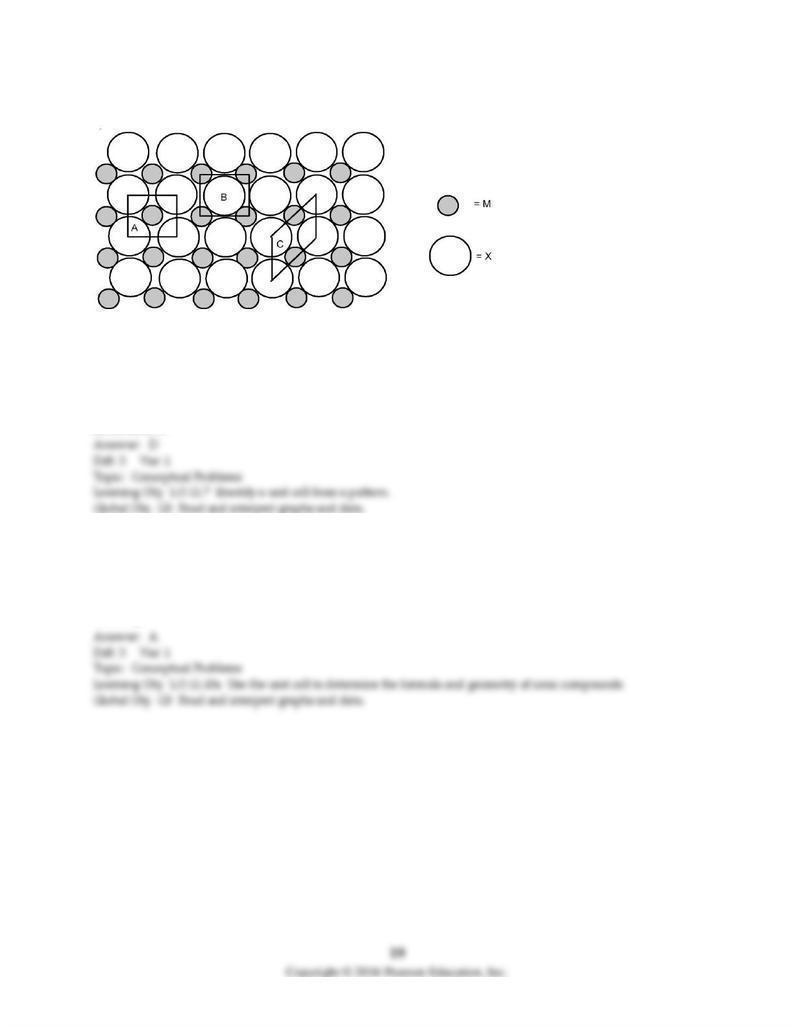
The picture shown below represents a two-dimensional lattice of atoms M and X.
62) Which of the areas designated A, B, and C are smallest repeating units, analogous to two-dimensional
unit cells?
A) only A
B) only B
C) only C
D) A, B, or C
63) What is the formula of the compound formed from M and X?
A) MX
B) MX2
C) MX3
D) MX4

64) Identify the packing in the figure shown below.
A) body-centered cubic
B) cubic closest packed (face-centered cubic)
C) hexagonal closest packed
D) simple cubic
65) Identify the packing in the figure shown below.
A) body-centered cubic
B) cubic closest packed (face-centered cubic)
C) hexagonal closest packed
D) simple cubic
66) Identify the packing in the figure shown below.
A) body-centered cubic
B) cubic closest packed (face-centered cubic)
C) hexagonal closest packed
D) simple cubic

67) Identify the packing in the figure shown below.
A) body-centered cubic
B) cubic closest packed (face-centered cubic)
C) hexagonal closest packed
D) simple cubic
68) What kind of packing do the anions adopt?
A) body-centered cubic
B) cubic closest packed (face-centered cubic)
C) hexagonal closest packed
D) simple cubic
69) How many cations and how many anions are in the unit cell?
A) 4 cations and 4 anions
B) 4 cations and 8 anions
C) 4 cations and 14 anions
D) 8 cations and 4 anions

70) A certain mineral crystallizes in the cubic unit cell shown below. M represents the cations and A
represents the anions. What is the empirical formula of the mineral?
A) MA
B) MA2
C) M2A
D) M4A4
71) How many cations M are in the unit cell?
A) 1
B) 2
C) 4
D) 8

72) How many anions are in the unit cell?
A) 1
B) 2
C) 3
D) 6
73) What is the empirical formula of the mineral?
A) MM'A
B) MM'A3
C) M2M'A3
D) M8M'A6
74) If cation M has a 2+ charge and anion A has a 2- charge, what is the oxidation state of cation M'?
A) +1
B) +2
C) +3
D) +4

75) Consider a compound that undergoes sublimation at 125°C and a pressure of one atm. Which of the
following could be a heating curve appropriate for heating the compound from 100°C to 150°C?
A) graph a
B) graph b
C) graph c
D) graph d
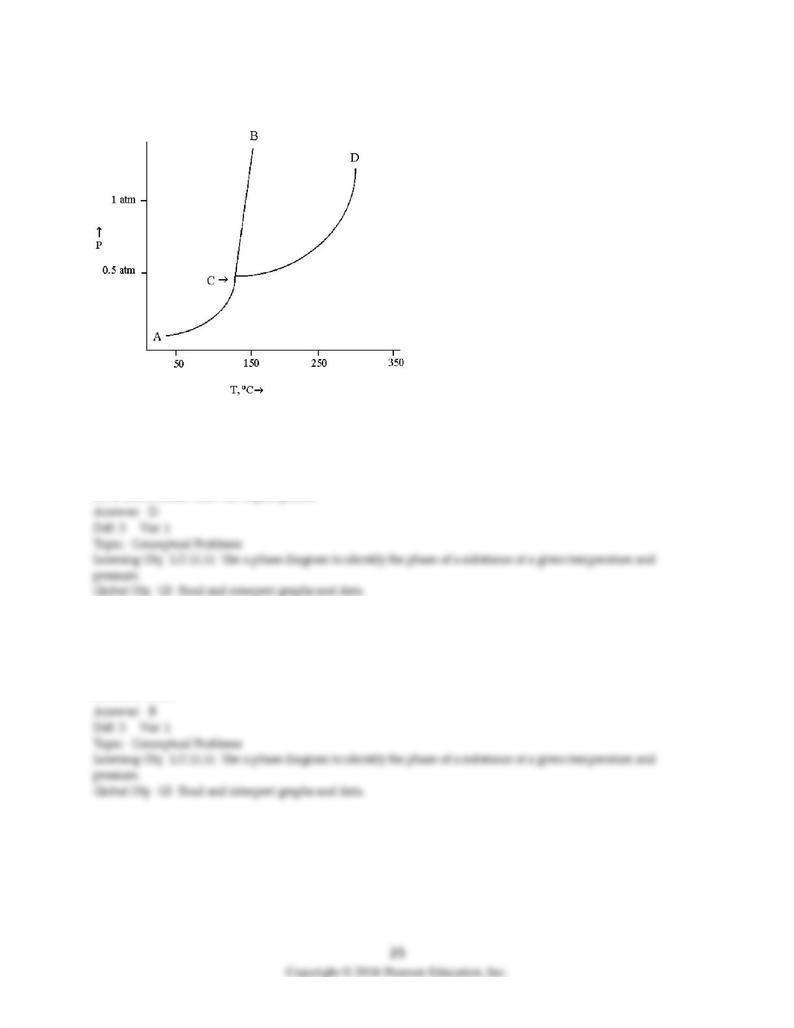
Use the diagram below to answer the following questions.
76) According to the diagram, the solid phase of this substance
A) has the same density as the liquid phase.
B) is less dense than water.
C) is less dense than the liquid phase.
D) is more dense than the liquid phase.
77) Melting occurs along the
A) AC line.
B) CB line.
C) CD line.
D) All of these

78) The solid and liquid phases can exist in equilibrium along line
A) AC.
B) CB.
C) CD.
D) BD.
79) The temperature and pressure at which all three phases can coexist in equilibrium is
A) 0.25 atm and 110°C.
B) 1.0 atm and 140°C.
C) 1.25 atm and 300°C.
D) 0.45 atm and 130°C.
80) From the phase diagram above, the minimum pressure at which this substance can exist in the liquid
phase is
A) 0.25 atm.
B) 0.45 atm.
C) 1.0 atm.
D) 1.2 atm.
81) The normal boiling point of this substance is approximately
A) 25°C.
B) 140°C.
C) 300°C.
D) 350°C.

82) What phases can be present at 200°C and 0.75 atm pressure?
A) only the vapor phase
B) only the liquid phase
C) only the solid phase
D) both the solid and vapor phases
83) What phase changes occur when the temperature is held constant at 140°C and the pressure is
increased from 0.25 atm to 1.4 atm?
A) gas → liquid → solid
B) gas → solid → liquid
C) liquid → solid → gas
D) solid → liquid → vapor
84) What phase changes occur when the pressure is held constant at 0.25 atm and the temperature
increases from 100°C to 300°C?
A) liquid → gas
B) solid → gas
C) solid → liquid
D) solid → liquid → gas

The phase diagram of a substance is shown below.
85) The approximate normal boiling point of this substance is
A) 180 K.
B) 190 K.
C) 300 K.
D) 430 K.
86) The approximate normal melting point of this substance is
A) 100 K.
B) 190 K.
C) 300 K.
D) 430 K.
87) What is the physical phase of the substance at T = 100 K and P = 0.1 atm?
A) gas
B) liquid
C) solid
D) supercritical fluid

88) What is the physical phase of the substance at T = 225 K and P = 1.1 atm?
A) gas
B) liquid
C) solid
D) supercritical fluid
89) What is the physical phase of the substance at T = 400 K and P = 2.0 atm?
A) gas
B) liquid
C) solid
D) supercritical fluid

11.2 Algorithmic Questions
1) The property of a liquid that measure the liquid's resistance to spread out and increase its surface area
is
A) sublimation point.
B) heat of deposition.
C) dipole-dipole forces.
D) surface tension.
2) Which of the following is most likely to have the highest viscosity at 25°C?
A) C6H14
B) HOCH2CH2OH
C) C3H7OH
D) C7H8
3) Which is expected to have the highest surface tension at 25°?
A) C7H16
B) C8H10
C) C5H11OH
D) C3H5(OH)3

4) Ethyl chloride, C2H5Cl, is used as a local anesthetic. It works by cooling tissue as it vaporizes; its heat
of vaporization is 26.4 kJ/mol. How much heat could be removed by 10.0 g of ethyl chloride?
A) 4.09 kJ
B) 170 kJ
C) 264 kJ
D) 1700 kJ
5) How much heat is released when 95.0 g of steam at 100.0°C is cooled to ice at -15.0°C? The enthalpy of
vaporization of water is 40.67 kJ/mol, the enthalpy of fusion for water is 6.01 kJ/mol, the molar heat
capacity of liquid water is 75.4 J/(mol ∙ °C), and the molar heat capacity of ice is 36.4 J/(mol ∙ °C).
A) 54.8 kJ
B) 247 kJ
C) 289 kJ
D) 314 kJ
6) Which of the following phase changes has a positive value for its entropy change?
A) creating steam
B) formation of snow from a cloud
C) sublimation of gaseous CO2
D) making ice cubes from liquid water
7) For which of the following phase changes is the sign of ΔS negative?
A) evaporation of water
B) formation of raindrops from water vapor in clouds
C) melting of ice cream
D) sublimation of CO2

8) When a liquid is heated at its boiling point, the
A) ionic bonds are broken, allowing sublimation to occur.
B) temperature of the liquid decreases.
C) temperature of the liquid remains the same as long as any liquid is present.
D) temperature of the vapor phase decreases.
9) The boiling point of a liquid is 64 °C and the enthalpy change for the conversion of this liquid to the
gas is 32.21 kJ/mole. What is the entropy change for vaporization, ΔSvap?
A) 2061 J/mol*K
B) 10.9 J/mol*K
C) 503 J/mol*K
D) 96 J/mol*K
10) How much heat (kJ) is required to convert 3.12 moles of liquid benzene at 75.1°C to gaseous benzene
at 115.1°C? The following information may be useful.
b.p = 80.1ᴼC
Cm (liquid benzene) = 136.0 J/ (mol * °C)
ΔHvap = 30.72 kJ/mol
Cm (gaseous benzene) = 36.6 J/ (mol * °C)
A) 94.0
B) 101965
C) 36.8
D) 102.0
11) Which of the following compounds has the highest boiling point?
A) H2O
B) HCl
C) N2
D) Ar

12) Which of the following substances has the highest boiling point?
A) CH3-CH2-CH2-CH2-CH3
B) Ar
C) CH3-CH2-CH2-CH3
D) (CH3)2CHCH2CH3
13) Arrange the following in order of increasing boiling point.
CH3CH2OH CH3CH2CH2CH3 H3C-O-CH2CH3 CH3CH2NH2
I II III IV
A) IV < III < II < I
B) II < III < IV < I
C) I < IV < III < II
D) II < III < I < IV
14) A kitchen pressure cooker operates at 1.70 atm. The ΔHvap of water is 40.7 kJ/mol. What is the boiling
point of water in the pressure cooker?
A) 380 K
B) 373 K
C) 389 K
D) 400 K
15) Which of the following forms a molecular solid?
A) CaCl2
B) C9H8O4
C) C, graphite
D) palladium

16) Which of the following forms an ionic solid?
A) Au
B) CH3NH2
C) NH4Cl
D) N H3
17) Which type of bonding does Ca form upon solidification?
A) covalent network
B) ionic
C) metallic
D) molecular
18) Solids having no ordered long-range structure are classified as
A) amorphous solids.
B) crystalline solids.
C) nonmetallic solids.
D) covalent network solids.
19) Which is classified as an amorphous solid?
A) palladium(II) bromide
B) phosphorus tetrachloride
C) plastic
D) potassium iodide

20) Which of the following forms a molecular solid?
A) lithium chloride
B) water
C) graphite
D) gold
21) Which of the following forms a metallic solid?
A) gold
B) potassium bromide
C) quartz
D) plastic
22) Which of the following compounds forms a covalent network solid?
A) Na
B) diamond
C) N2
D) CS2
23) Which of the following compounds forms an ionic solid?
A) water
B) carbon dioxide
C) potassium bromide
D) zince

24) Which type of bonding does sucrose form upon solidification?
A) metallic
B) amorphous
C) molecular
D) anionic
25) What is the edge length of a face-centered cubic unit cell made up of atoms having a radius of 139
pm?
A) 197 pm
B) 393 pm
C) 556 pm
D) 1110 pm
26) Aluminum has a face-centered cubic structure and has a density of 2.70 g/cm3. What is its atomic
radius?
A) 143 pm
B) 286 pm
C) 405 pm
D) 1150 pm
27) A certain metal crystallizes in a face-centered cubic structure. What is the edge length of the unit cell if
the atomic radius of the metal is 134 pm?
A) 189 pm
B) 268 pm
C) 309 pm
D) 379 pm

28) Barium has a radius of 224 pm and crystallizes in a body-centered cubic structure. What is the edge
length of the unit cell?
A) 259 pm
B) 317 pm
C) 448 pm
D) 517 pm
29) Manganese crystallizes in a body-centered cubic structure. What is the coordination number of each
atom?
A) 4
B) 6
C) 8
D) 12
30) KI crystallizes in a cubic unit cell with I- ions on each corner and each face. How many K+ and I- ions
are in each unit cell of KI?
A) 1 K+ ion and 1 I- ion
B) 2 K+ ions and 2 I- ions
C) 4 K+ ions and 4 I- ions
D) 8 K+ ions and 8 I- ions

31) How many Br- ions are around each Na+ ion in NaBr, which has a cubic unit cell with Br- ions on
each corner and each face?
A) 1
B) 4
C) 6
D) 8
32) Which of the following is an allotrope of carbon?
A) diamond
B) glass
C) coal
D) carbon monoxide
33) The layers of graphite are held together by
A) covalent bonds.
B) ion-ion forces.
C) London dispersion forces.
D) cationic forces.
34) Diamond is held together by
A) covalent bonds.
B) ion-dipole forces.
C) van der Waals forces.
D) ionic forces.

35) ________ are used as a battery electrode material.
A) Buckyballs
B) Graphite
C) Carbon
D) Sucrose molecules
36) Which transition could occur if a solid is heated at a pressure above the triple point pressure?
A) vaporization
B) deposition
C) melting
D) sublimation
11.3 Short Answer Questions
1) The property of a liquid that is a measure of the liquid's resistance to increase its surface area is
________.
2) Of C2H5OH and C3H5(OH)3 the one expected to have the higher viscosity is ________, and the one
expected to have the higher surface tension is ________.
3) The phase change H2(g) → H2(s) is called ________, and the enthalpy change, ΔH, for this phase
change has a ________ sign.

4) Of C2H5OH and C3H5(OH)3 the one expected to have the higher vapor pressure is ________, and the
one expected to have the higher boiling point is ________.
5) The solids formed by Na, Na2O2, SiO2, and N2 are classified as ________, ________, ________, and
________, respectively.
6) Rubber is classified as an ________ solid, whereas diamond is classified as a ________ solid.
7) A low-melting crystalline compound that does not conduct electricity in the solid or liquid state is
classified as a ________ solid.
8) First-order diffraction of X-rays with d = 154.2 pm at an angle of 32.5° is caused by layers of atoms in a
crystalline solid with a spacing of ________ pm.
9) Layers of atoms having a spacing of 105 pm will diffract X-rays with d = 154.2 pm at an angle of
________ degrees.

10) The cubic closest-packed arrangement of atoms is the same as which cubic unit cell?
11) The two most efficiently packed unit cells have the hexagonal closest-packed and the ________ the
atomic arrangements.
12) The coordination number of each atom in a body-centered cubic unit cell is ________.
13) The cubic unit cell in which the radius of an atom is 31/2 d/4, where d is the unit cell edge length, is
the ________ unit cell.
14) A certain metal can exist in two different cubic cells, body-centered and face-centered cubic. Which
unit cell will have the greater density?
15) Ni has a face-centered unit cell. The number of Ni atoms in the unit cell is ________.

16) A compound having A ions on each corner and B ions on each face of a cubic unit cell has the
empirical formula ________.