
Chemistry, 7e (McMurry/Fay)
Chapter 12 Solutions and Their Properties
12.1 Multiple-Choice Questions
1) Which of the following mixtures have components which can be separated by filtration?
A) colloids
B) solutions
C) suspensions
D) all of these
2) In most liquid solutions, the component present in the larger amount is called the
A) dispersed medium.
B) emulsifying agent.
C) solute.
D) solvent.
3) The rubbing alcohol sold in drug stores often is composed of 70% isopropyl alcohol and 30% water. In
this solution
A) isopropyl alcohol is the solvent.
B) water is the solvent.
C) both water and isopropyl alcohol are solvents.
D) neither water nor isopropyl alcohol is a solvent.
4) For a liquid solution made by dissolving a solid or a gas in a liquid, the
A) liquid is the solute.
B) liquid is the solvent.
C) solute is the component present in the greatest amount.
D) solvent is the component present in the greatest amount.

5) Iodine, I2(s), is more soluble in dichloromethane, CH2Cl2(l), than in water because
A) both iodine and dichloromethane have strong ion-dipole interactions.
B) the dipole-dipole forces in dichloromethane are much stronger than the dispersion forces in iodine.
C) the intermolecular forces are similar in both iodine and dichloromethane.
D) iodine is polar and dichloromethane has a large number of hydrogen bonds.
6) For which case would ΔHsoln be expected to be negative?
A) if solute-solute interactions are much greater than solvent-solvent and solute-solvent interactions
B) if solvent-solvent interactions are much greater than solute-solvent and solute-solute interactions
C) if solute-solvent interactions are much greater than solvent-solvent and solute-solute interactions
D) if solute-solvent interactions are the same as solvent-solvent and solute-solute interactions
7) Commercial cold packs often contain solid NH4NO3 and a pouch of water. The temperature of the
pack drops as the NH4NO3 dissolves in water. Therefore, for the dissolving of NH4NO3 in water,
A) ΔHsoln is negative and ΔSsoln may be negative or positive.
B) ΔHsoln is negative and ΔSsoln is positive.
C) ΔHsoln is positive and ΔSsoln may be negative or positive.
D) ΔHsoln is positive and ΔSsoln is positive.
8) When a particular solid begins to dissolve in water, the temperature rises dramatically. For the
dissolving of this solid in pure water
A) ΔHsoln is always negative and ΔSsoln may be negative or positive.
B) ΔHsoln is always negative and ΔSsoln is always positive.
C) ΔHsoln is always positive and ΔSsoln may be negative or positive.
D) ΔHsoln is always positive and ΔSsoln is always positive.

9) One reason ionic compounds do not dissolve well in nonpolar solvents is that
A) ion-dipole interactions are too large for effective solvation to occur.
B) ion-solvent interactions are not strong enough to solvate the ions in solution.
C) not all cations and anions have the same magnitude of charge and therefore do not form neutral ion
pairs.
D) there are no forces of attraction between ions and nonpolar molecules.
10) Which cation in each set is expected to have the larger (more negative) hydration energy?
I. Mg2+ or Ca2+
II. Li+ or Al3+
A) Mg2+ in set I and Li+ in set II
B) Mg2+ in set I and Al3+ in set II
C) Ca2+ in set I and Li+ in set II
D) Ca2+ in set I and Al3+ in set II
11) Although there are exceptions, which is most likely to be true for the dissolving of a solid in a liquid?
A) ΔHsoln is positive.
B) ΔHsoln is negative.
C) ΔSsoln is positive.
D) ΔSsoln is negative.
12) When two similar liquids mix to form a solution, the entropy of solution (ΔSsoln) is expected to be
A) negative.
B) zero.
C) positive.
D) negative at low temperatures but positive at high temperatures.

13) When an ionic solute dissolves in water to form an unsaturated solution, the free energy change
(ΔGsoln) is
A) negative.
B) zero.
C) positive.
D) either A or C, depending on the ionic compound.
14) The change in the Gibbs free energy for dissolving more solute in a supersaturated solution is
A) negative.
B) zero.
C) positive.
D) positive at low temperatures and negative at high temperatures.
15) The change in the Gibbs free energy for dissolving solute in a saturated solution is
A) negative.
B) zero.
C) positive.
D) positive at low temperatures and negative at high temperatures.
16) For the process of dissolving a solid in a liquid, which of the following statements is true?
A) ΔHsoln is always negative and ΔSsoln is usually positive.
B) ΔHsoln is always positive and ΔSsoln is usually negative.
C) ΔHsoln is either positive or negative and ΔSsoln is usually positive.
D) ΔHsoln is either positive or negative and ΔSsoln is usually negative.

17) Most gases become less soluble in water as the temperature increases. What can be concluded about
the signs of ΔHsoln and ΔSsoln in this case?
A) ΔHsoln is negative and ΔSsoln is negative.
B) ΔHsoln is negative and ΔSsoln is positive.
C) ΔHsoln is positive and ΔSsoln is negative.
D) ΔHsoln is positive and ΔSsoln is positive.
18) Which of the following should most favor the solubility of an ionic solid in water?
A) a low lattice energy for the solid and a low hydration energy for its ions
B) a low lattice energy for the solid and a high hydration energy for its ions
C) a high lattice energy for the solid and a low hydration energy for its ions
D) a high lattice energy for the solid and a high hydration energy for its ions

19) Which should be least soluble in water?
A)
B)
C)
D)
20) Formaldehyde is a carcinogenic volatile organic compound with a permissible exposure level of 0.75
ppm. At this level, how many grams of formaldehyde are permissible in a 6.0-L breath of air having a
density of 1.2 kg/m3?
A) 3.8 × 10-2 g formaldehyde
B) 5.4 × 10-6 g formaldehyde
C) 3.8 g formaldehyde
D) 5.4 g formaldehyde

21) What is the mole fraction of ethanol in a solution made by dissolving 25.0 g of ethanol, C2H5OH, in
53.6 g of water?
A) 0.154
B) 0.846
C) 0.214
D) 0.272
22) What is the mole fraction of I2 in a solution made by dissolving 27.8 g of I2 in 260 g of hexane, C6H14?
A) 0.0350
B) 0.965
C) 1.03
D) 28.57
23) What is the mole fraction of oxygen in a gas mixture that is 40.0% oxygen and 60.0% nitrogen by
volume?
A) 0.632
B) 0.368
C) 0.400
D) 0.600
24) A solution is prepared by dissolving 17.75 g sulfuric acid, H2SO4, in enough water to make 100.0 mL
of solution. If the density of the solution is 1.1094 g/mL, what is the mole fraction H2SO4 in the solution?
A) 0.0181
B) 0.0338
C) 0.0350
D) 19.0

25) A solution is prepared by dissolving 17.75 g sulfuric acid, H2SO4, in enough water to make 100.0 mL
of solution. If the density of the solution is 1.1094 g/mL, what is the molarity?
A) 0.1775 M H2SO4
B) 0.1810 M H2SO4
C) 1.810 M H2SO4
D) 1.940 M H2SO4
26) A solution is prepared by dissolving 17.75 g sulfuric acid, H2SO4, in enough water to make 100.0 mL
of solution. If the density of the solution is 1.1094 g/mL, what is the molality?
A) 0.1775 m H2SO4
B) 0.1810 m H2SO4
C) 1.810 m H2SO4
D) 1.940 m H2SO4
27) A solution is prepared by dissolving 17.75 g sulfuric acid, H2SO4, in enough water to make exactly
100.0 mL of solution. If the density of the solution is 1.1094 g/mL, what is the weight % H2SO4 in the
solution?
A) 16.00%
B) 18.00%
C) 19.00%
D) 84.00%
28) A solution is prepared by dissolving 171 g of CdCl2 in enough water to make 250.0 mL of solution. If
the density of the solution is 1.556 g/mL, what is the molarity of the solution?
A) 0.440 M
B) 0.684 M
C) 0.933 M
D) 3.73 M

29) A solution is prepared by dissolving 171 g of CdCl2 in enough water to make exactly 250.0 mL of
solution. If the density of the solution is 1.556 g/mL, what is the weight percent of CdCl2 in the solution?
A) 7.17%
B) 44.0%
C) 56.0%
D) 68.4%
30) What is the weight percent of vitamin C in a solution made by dissolving 8.00 g of vitamin C,
C6H8O6, in 55.0 g of water?
A) 87.3%
B) 14.5%
C) 12.7%
D) 85.5%
31) What is the weight percent of a caffeine solution made by dissolving 4.00 g of caffeine, C8H10N4O2,
in 75.0 g of benzene, C6H6?
A) 0.0506%
B) 0.0533%
C) 5.06%
D) 5.33%
32) What volume of 0.716 M KBr solution is needed to provide 13.0 g of KBr?
A) 6.55 mL
B) 9.31 mL
C) 18.5 mL
D) 153 mL

33) What volume of 3.00 M CH3OH solution is needed to provide 0.500 mol of CH3OH?
A) 3.12 mL
B) 9.38 mL
C) 167 mL
D) 150 mL
34) How much water must be added to 42.0 g of CaCl2 to produce a solution that is 40.0 wt% CaCl2?
A) 56.7 g
B) 63.0 g
C) 16.8 g
D) 120 g
35) To make a 3.0 M solution, one could take 3.00 moles of solute and add
A) 1.00 L of solvent.
B) 1.00 kg of solvent.
C) enough solvent to make 1.00 L of solution.
D) enough solvent to make 1.00 kg of solution.
36) To make a 3.00 m solution, one could take 3.00 moles of solute and add
A) 1.00 L of solvent.
B) 1.00 kg of solvent.
C) enough solvent to make 1.00 L of solution.
D) enough solvent to make 1.00 kg of solution.

37) What molality of pentane is obtained by dissolving 50.0 g pentane, C5H12, in 245.0 g hexane, C6H14?
A) 0.200 m
B) 0.240 m
C) 2.83 m
D) 200. m
38) A solution is 4.50% by weight NaHCO3. How many grams of NaHCO3 are in 450.0 g of solution?
A) 1.000 g
B) 20.2 g
C) 400 g
D) 450 g
39) How many grams of KBr are required to make 850. mL of a 0.115 M KBr solution?
A) 0.0978 g
B) 85.9 g
C) 11.6 g
D) 13.7 g
40) What is the molality of a glucose solution prepared by dissolving 9.00 g of glucose, C6H12O6, in 125.9
g of water?
A) 2.38 × 10-4 m
B) 0.0715 m
C) 0.347 m
D) 0.397 m

41) Aqueous solutions of 30% (by weight) hydrogen peroxide, H2O2, are used to oxidize metals or
organic molecules in chemical reactions. Given that the density of the solution is 1.11 g/mL, calculate the
molarity.
A) 0.794 M
B) 6.78 M
C) 9.79 M
D) 12.6 M
42) Aqueous solutions of 30.0% (by weight) hydrogen peroxide, H2O2, are used to oxidize metals or
organic molecules in chemical reactions. Calculate the molality of this solution.
A) 0.974 m
B) 6.78 m
C) 9.79 m
D) 12.6 m
43) Sodium hydroxide is available commercially as a 50.0% by weight aqueous solution. The density of
the solution is 1.53 g/mL. Calculate the molarity of this sodium hydroxide solution.
A) 0.450 M
B) 19.1 M
C) 25.0 M
D) 125. M
44) Sodium hydroxide is available commercially as a 50.0% by weight aqueous solution. Calculate the
molality of this sodium hydroxide solution.
A) 0.450 m
B) 19.1 m
C) 25.0 m
D) 125. m

45) A 3.17 m solution of CaCl2 in water has a density of 1.24 g/mL. What is the molarity?
A) 2.56 M CaCl2
B) 2.91 M CaCl2
C) 3.50 M CaCl2
D) 3.93 M CaCl2
46) A 2.00 M solution of CaCl2 in water has a density of 1.17 g/mL. What is the mole fraction of CaCl2?
A) 0.0348
B) 0.0360
C) 0.0366
D) 0.0380
47) A solution of LiCl in water has XLiCl = 0.0400. What is the molality?
A) 4.16 m LiCl
B) 2.22 m LiCl
C) 2.31 m LiCl
D) 4.21 m LiCl
48) A solution of LiCl in water is 9.00 wt% LiCl. What is the mole fraction of LiCl?
A) 0.0407
B) 0.0466
C) 0.212
D) 2.28

49) At 25.0°C, a solution has a concentration of 3.179 M and a density of 1.260 g/mL. The density of the
solution at 50.0°C is 1.249 g/mL. What is the molarity of the solution at 50.0°C?
A) 2.545 M
B) 3.151 M
C) 3.179 M
D) 3.230 M
50) Which of the following aqueous salt (NaCl) solutions has the greater concentration?
A) 0.5 M
B) 2.3 m
C) 3.0 % by mass
D) 11.1 g/L
51) Which of the following statements is true for a supersaturated solution?
A) The solute in the solution is at equilibrium with undissolved solute.
B) The solution contains more than the equilibrium amount of solute.
C) The solution is stable and the solute will not precipitate.
D) A supersaturated solution is more than 50% solute by mass.
52) In general, as the temperature increases, the solubility of gases in water ________ and the solubility of
most solids in water ________.
A) decreases, decreases
B) decreases, increases
C) increases, decreases
D) increases, increases

53) The solubility of gaseous solutes in liquid solvents is greater when the
A) external pressure over the solution is increased.
B) external pressure is decreased.
C) partial pressure of the gas above the solution is increased.
D) partial pressure of the solvent is increased.
54) The Henry's Law constant of methyl bromide, CH3Br, is k = 0.159 mol/(L ∙ atm) at 25°C. What is the
solubility of methyl bromide in water at 25°C and at a partial pressure of 250. mm Hg?
A) 0.0523 mol/L
B) 0.329 mol/L
C) 0.483 mol/L
D) 39.8 mol/L
55) The solubility of argon in water at 25°C is 0.0150 mol/L. What is the Henry's Law constant for argon if
the partial pressure of argon in air is 0.00934 atm?
A) 1.40 × 10-4 mol/(L ∙ atm)
B) 0.623 mol/(L ∙ atm)
C) 1.61 mol/(L ∙ atm)
D) 4.10 mol/(L ∙ atm)
56) Which of the following is not an application of colligative properties?
A) adding silver to mercury to lower the vapor pressure of mercury
B) desalinating sea water by reverse osmosis
C) melting snow by application of salt
D) reduced boiling points of pure liquids at increased altitudes

57) A solution is prepared by dissolving 20.0 g of sucrose, C12H22O11, in 250. g of water at 25°C. What is
the vapor pressure of the solution if the vapor pressure of water at 25°C is 23.76 mm Hg?
A) 0.198 mm Hg
B) 20.5 mm Hg
C) 23.7 mm Hg
D) 24.0 mm Hg
58) A KCl solution is prepared by dissolving 60.0 g KCl in 250.0 g of water at 25°C. What is the vapor
pressure of the solution if the vapor pressure of water at 25°C is 23.76 mm Hg?
A) 20.5 mm Hg
B) 21.3 mm Hg
C) 22.5 mm Hg
D) 25.5 mm Hg
59) At a given temperature the vapor pressures of benzene and toluene are 183 mm Hg and 59.2 mm Hg,
respectively. Calculate the total vapor pressure over a solution of benzene and toluene with Xbenzene =
0.400.
A) 110 mm Hg
B) 133 mm Hg
C) 109 mm Hg
D) 242 mm Hg
60) At a given temperature the vapor pressures of benzene and toluene are 183 mm Hg and 59.2 mm Hg,
respectively. Calculate the mole fraction of benzene in the vapor phase over a solution of benzene and
toluene with Xbenzene = 0.600.
A) 0.600
B) 0.678
C) 0.756
D) 0.823

61) How many grams of sucrose, C12H22O11, must be added to 500. g of water at 100°C to change the
vapor pressure to 752 mm Hg?
A) 0.295 g
B) 5.32 g
C) 10.6 g
D) 101 g
62) At 25°C the vapor pressures of benzene and toluene are 96.0 mm Hg and 30.5 mm Hg, respectively.
When a 1:1 molar mixture of benzene and toluene is fractionally distilled, the first fraction will have a
mole fraction of benzene that is closest to
A) 0.0.
B) 0.5.
C) 0.7.
D) 1.0.
63) Which of the following solutions will have the lowest freezing point?
A) 0.0100 m NaCl
B) 0.0120 m Li2SO4
C) 0.0400 m CH3CH2CH2OH
D) 0.0150 m MgCl2
64) What is the expected freezing point of a 0.50 m solution of Na2CO3 in water? Kf for water is 1.86°C/m.
A) -0.93°C
B) -1.9°C
C) -2.8°C
D) -6.5°C

65) Calculate the freezing point of a solution of 50.0 g methyl salicylate, C7H6O2, dissolved in 800. g of
benzene, C6H6. Kf for benzene is 5.10°C/m and the freezing point is 5.50°C for benzene.
A) -2.61°C
B) 2.61°C
C) 2.89°C
D) 8.39°C
66) What is the freezing point of a solution of 8.00 g MgCl2 in 100 g of water? Kf for water is 1.86°C/m for
water.
A) 1.56°C
B) -1.56°C
C) 4.69°C
D) -4.69°C
67) The normal boiling point of pure benzene is found to be 80.10°C. What is the approximate molecular
weight of a nonionizing substance if a solution of 3.55 g of the substance dissolved in 100. g of benzene
has a normal boiling point of 80.19°C? Kb = 5.12°C/m for benzene, C6H6.
A) 20 amu
B) 500 amu
C) 2000 amu
D) 20,000 amu
68) An aqueous solution has a normal boiling point of 105.0°C. What is the freezing point of this solution?
For water Kb is 0.51°C/m and Kf = 1.86°C/m.
A) 14.7°C
B) -14.7°C
C) 18.2°C
D) -18.2°C

69) A solution of 0.2113 g of water dissolved in 25.0 g of a solvent freezes at 11.5°C below the freezing
point of the solvent. What is Kf for this solvent?
A) 0.735°C/m
B) 1.36°C/m
C) 5.39°C/m
D) 24.5°C/m
70) An aqueous CsCl solution is 8.00 wt% CsCl and has a density of 1.0643 g/mL at 20°C. What is the
boiling point of this solution? Kb = 0.51°C/m for water.
A) 100.27°C
B) 100.53°C
C) 103.8°C
D) 104.3°C
71) When 0.500 g of vitamin K is dissolved in 10.0 g of camphor (Kf = 40.0°C/m), the freezing point of the
solution is 4.43°C lower than that of pure camphor. Assuming vitamin K is a nonelectrolyte in camphor,
calculate its molar mass.
A) 0.451 g/mol
B) 55.4 g/mol
C) 451 g/mol
D) 3.54 × 104 g/mol
72) When ethylene glycol, HOCH2CH2OH, is added to the water in an automobile radiator, the effect is
to
A) lower the boiling point and lower the freezing point.
B) lower the boiling point and raise the freezing point.
C) raise the boiling point and lower the freezing point.
D) raise the boiling point and raise the freezing point.

73) The coolant in automobiles is often a 50/50 % by volume mixture of ethylene glycol, HOCH2CH2OH,
and water. At 20°C, the density of ethylene glycol is 1.1088 g/mL and the density of water is 0.9982 g/mL.
Assuming that the volumes are additive, what is the expected freezing point of a 50/50(v/v)% ethylene
glycol/water solution? Kf = 1.86°C/m for water.
A) -16°C
B) -17°C
C) -30°C
D) -33°C
74) Two aqueous solutions, A and B, are separated by a semipermeable membrane. The osmotic pressure
of solution A immediately begins to decrease. Which of the following statements is true?
A) Solvent molecules are moving from solution B into solution A.
B) The initial osmotic pressure of solution B is greater than that of solution A.
C) The solvent molecules are moving from the solution of higher osmotic pressure to that of lower
osmotic pressure.
D) Both B and C are true statements.
75) Red blood cells are placed into pure water. Which of the following statements is true?
A) Water molecules flow out of the red blood cells, causing them to collapse.
B) Water flows into the red blood cells, causing them to swell and burst.
C) The osmotic pressure of the cell contents increases, causing the cells to burst.
D) The osmotic pressure inside the cells equals the osmotic pressure outside.

76) How will the osmotic pressure of an aqueous solution change as evaporation occurs?
A) The osmotic pressure will increase.
B) The osmotic pressure will not change.
C) The osmotic pressure will decrease.
D) The osmotic pressure will increase or decrease until it equals the vapor pressure of water.
77) Assuming that sea water is a 3.5 wt % solution of NaCl in water, calculate its osmotic pressure at
20°C. The density of a 3.5% NaCl solution at 20°C is 1.023 g/mL.
A) 1.0 atm
B) 15 atm
C) 29 atm
D) 100 atm
78) The average osmotic pressure of blood is about 7 atm. Therefore
A) the average blood pressure is about 7 atm.
B) the average pressure inside the body is about 7 atm above the external pressure.
C) a pressure of about 7 atm would be required to prevent osmosis if blood is in contact with pure water
across a semipermeable membrane.
D) All of these are true.

79) Naproxen is a commercially important anti-inflammatory agent that can be isolated from the thyroid
gland. A solution of 1.138 g of naproxen in 25.00 g benzene has an osmotic pressure of 4.00 atm at 20°C.
The density of benzene is 0.8787 g/mL at this temperature. Calculate the molar mass of naproxen,
assuming it remains intact upon dissolution and the density of the solution equals the density of pure
benzene.
A) 176 g/mol
B) 230 g/mol
C) 307 g/mol
D) 3.80 × 105 g/mol
80) A solution of 62.4 g of insulin in enough water to make 1.000 L of solution has an osmotic pressure of
0.305 atm at 25°C. Based on these data, what is the molar mass of insulin?
A) 621 g/mol
B) 5000 g/mol
C) 7570 g/mol
D) 71,900 g/mol
81) A solution of a nonelectrolyte solution contains 30.0 g of solute dissolved in 250.0g of water. The
freezing point of the water is observed to be -2.50˚C. The Kf for water is 1.86 ˚C/m and normal freezing
point of water is 0.00˚C. What is the molar mass of the substance?
A) 335 g/mol
B) 89.5 g/mol
C) 895 g/mol
D) 33.5 g/mol

82) Which drawing above represents the system with the highest entropy?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
83) Which drawing above represents the system with the lowest entropy?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
84) Which drawing above represents the system with the second lowest entropy?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
85) Which drawing above represents the system with the second highest entropy?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)

Arrows in the energy diagram below represent enthalpy changes occurring in the exothermic formation
86) Which arrow represents ΔHsoln?
A) arrow (a)
B) arrow (b)
C) arrow (c)
D) arrow (d)
87) Which arrow represents ΔHsolute-solvent?
A) arrow (a)
B) arrow (b)
C) arrow (c)
D) arrow (d)

88) Which arrows represent ΔHsolute-solute and ΔHsolvent-solvent?
A) arrow (a) and arrow (b)
B) arrow (a) and arrow (c)
C) arrow (a) and arrow (d)
D) arrow (c) and arrow (d)
89) Which arrow represents ΔHsoln?
A) arrow (a)
B) arrow (b)
C) arrow (c)
D) arrow (d)
90) Which arrow represents ΔHsolute-solvent?
A) arrow (a)
B) arrow (b)
C) arrow (c)
D) arrow (d)
91) Which arrows represent ΔHsolute-solute and ΔHsolvent-solvent?
A) arrow (a) and arrow (b)
B) arrow (a) and arrow (c)
C) arrow (a) and arrow (d)
D) arrow (c) and arrow (d)
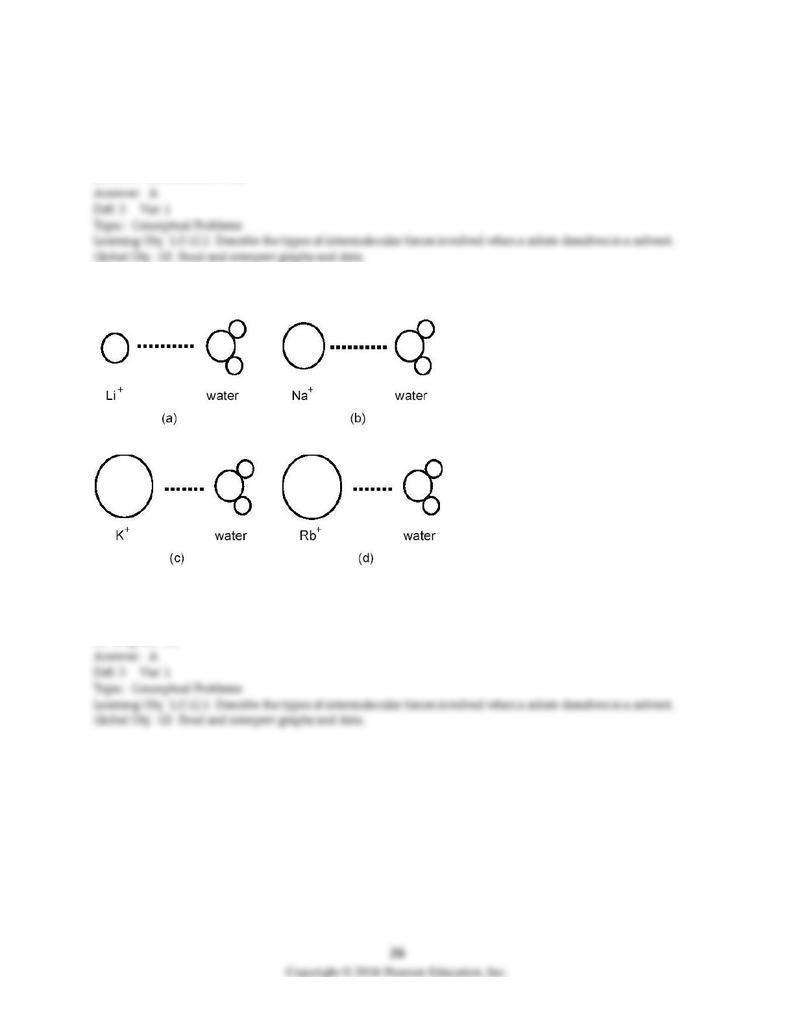
92) Which ion-dipole interaction results in the larger (more negative) hydration energy?
A) diagram (a)
B) diagram (b)
C) diagram (c)
D) diagram (d)
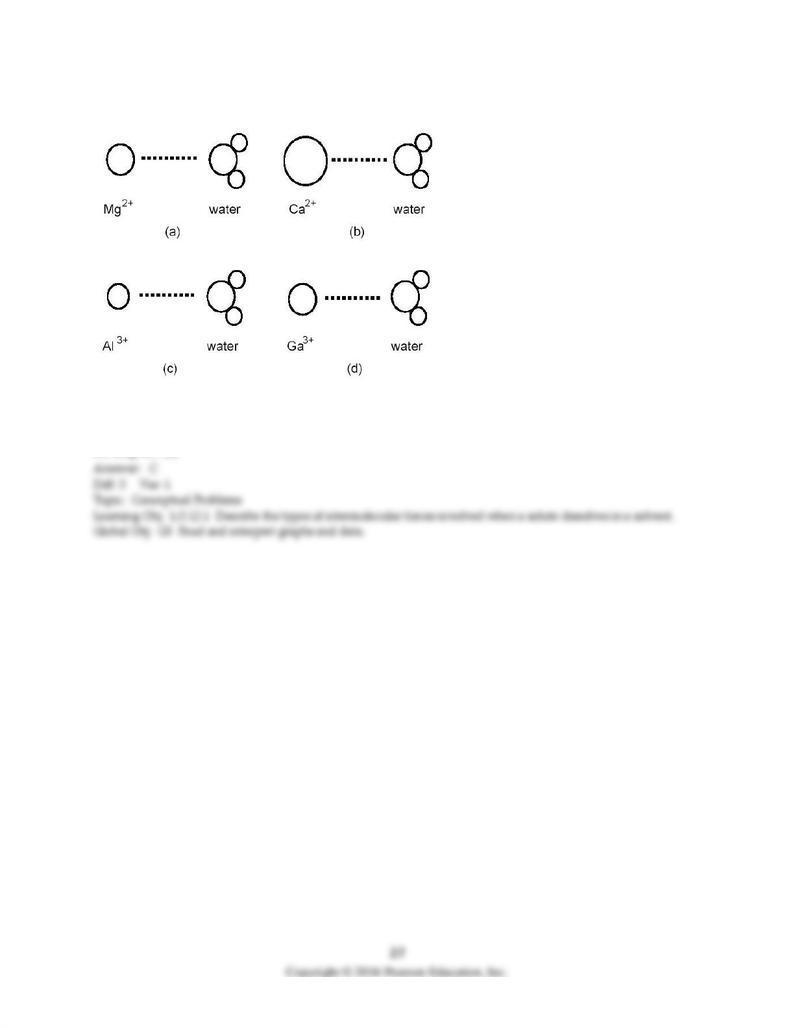
93) Which ion-dipole interaction results in the larger (more negative) hydration energy?
A) diagram (a)
B) diagram (b)
C) diagram (c)
D) diagram (d)

94) Drawing (1) shows a system in which an equilibrium exists between dissolved and undissolved gas
particles at P = 1 atm. According to Henry's law, if the pressure is increased to 2 atm and equilibrium is
restored, which drawing (2)-(5) best represents the equilibrium at 2 atm?
A) drawing (2)
B) drawing (3)
C) drawing (4)
D) drawing (5)

95) Drawing (1) shows a system in which an equilibrium exists between dissolved and undissolved gas
particles at P = 1 atm. According to Henry's law, if the pressure is decreased to 0.5 atm and equilibrium is
restored, which drawing (2)-(5) best represents the equilibrium at 0.5 atm?
A) drawing (2)
B) drawing (3)
C) drawing (4)
D) drawing (5)

96) Drawing (1) shows the equilibrium vapor pressure of a pure liquid. Which drawing (2)-(5) represents
the equilibrium vapor pressure when a nonvolatile solute is dissolved in the liquid?
A) drawing (2)
B) drawing (3)
C) drawing (4)
D) drawing (5)

97) Drawings (1) and (2) show the equilibrium vapor pressures of two pure liquids. Which drawing (3)-
(6) represents the equilibrium vapor pressure of a solution made by mixing equal moles of each liquid?
A) drawing (3)
B) drawing (4)
C) drawing (5)
D) drawing (6)

The following diagram shows a close-up view of the vapor pressure curves for a pure solvent and a
98) Which curve is the solvent and what happens to the vapor pressure when the solute is dissolved in
the solvent?
A) Curve (a) is the solvent and the vapor pressure decreases.
B) Curve (a) is the solvent and the vapor pressure increases.
C) Curve (b) is the solvent and the vapor pressure decreases.
D) Curve (b) is the solvent and the vapor pressure increases.
99) Which curve is the solvent and what happens to the boiling point when the solute is dissolved in the
solvent?
A) Curve (a) is the solvent and the boiling point decreases.
B) Curve (a) is the solvent and the boiling point increases.
C) Curve (b) is the solvent and the boiling point decreases.
D) Curve (b) is the solvent and the boiling point increases.

100) The following diagram shows a close-up view of the vapor pressure curves for two pure liquids and
two different solutions composed of these two liquids. Which curves represent pure liquids and which
curves represent the solutions?
A) Curves (a) and (b) are the pure liquids and curves (c) and (d) are the solutions.
B) Curves (a) and (c) are the pure liquids and curves (b) and (d) are the solutions.
C) Curves (a) and (d) are the pure liquids and curves (b) and (c) are the solutions.
D) Curves (c) and (d) are the pure liquids and curves (a) and (b) are the solutions.
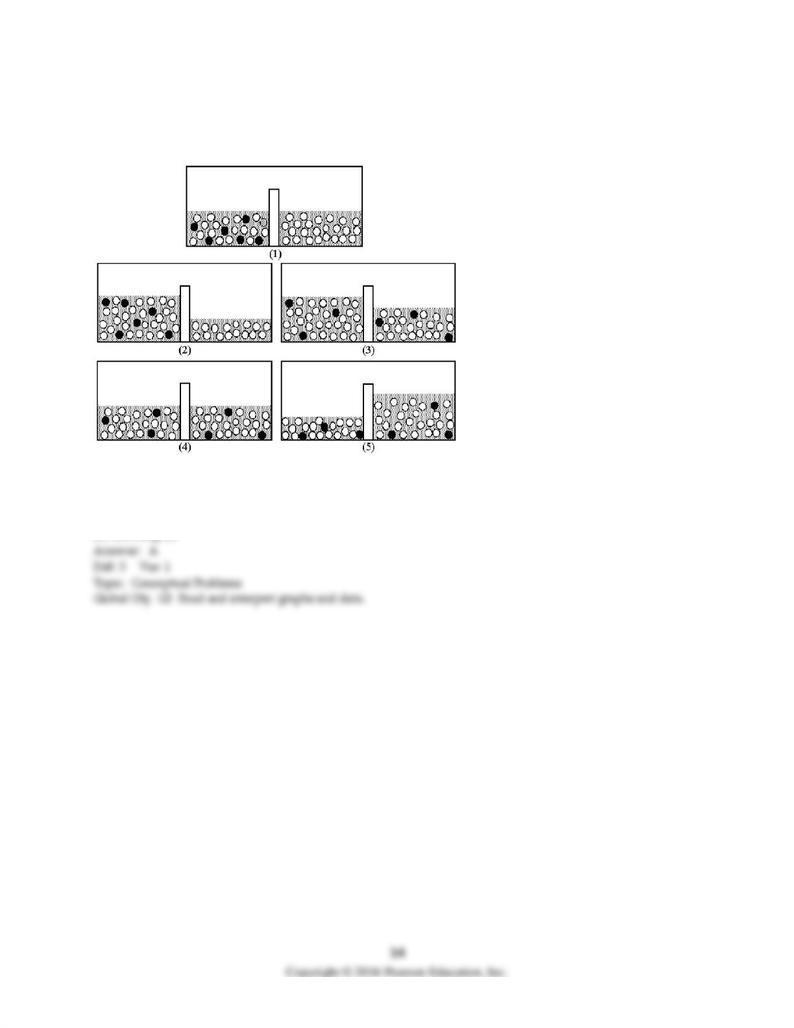
101) Drawing (1) shows a nonequilibrium system comprised of pure water separated from an aqueous
solution by a semipermeable membrane. Shaded spheres represent solute particles and unshaded spheres
represent water molecules. Which drawing (2)-(5) represents this system after equilibrium is reached?
A) drawing (2)
B) drawing (3)
C) drawing (4)
D) drawing (5)

102) Two beakers, one with pure water (light gray) and the other with an aqueous solution of KBr (dark
gray), are placed in a closed container represented by drawing (a). Which of the drawings (a)-(d)
represents what the beakers will look like after a substantial amount of time has passed?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
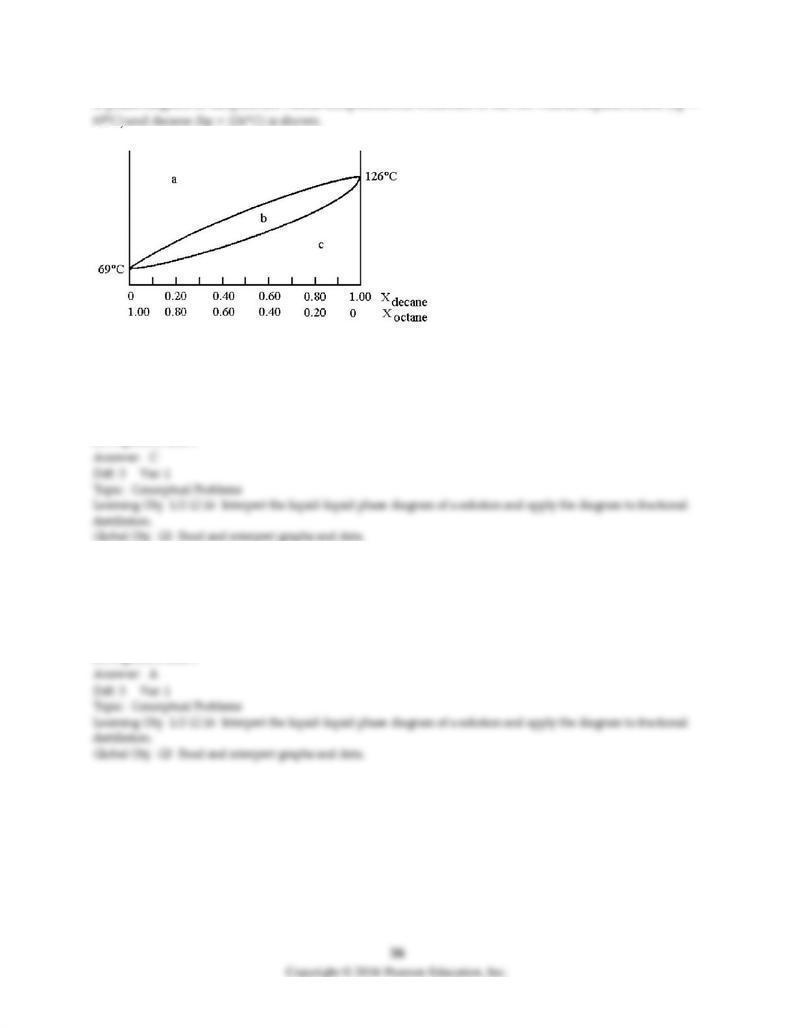
A phase diagram of temperature versus composition for a mixture of the two volatile liquids octane (bp =
103) Assume that you start with a mixture containing 0.80 mol of decane and 0.20 mol of octane, what
region of the diagram corresponds to liquid?
A) region a
B) region b
C) region c
D) regions a and c
104) Assume that you start with a mixture containing 0.80 mol of decane and 0.20 mol of octane, what
region of the diagram corresponds to vapor?
A) region a
B) region b
C) region c
D) regions a and c
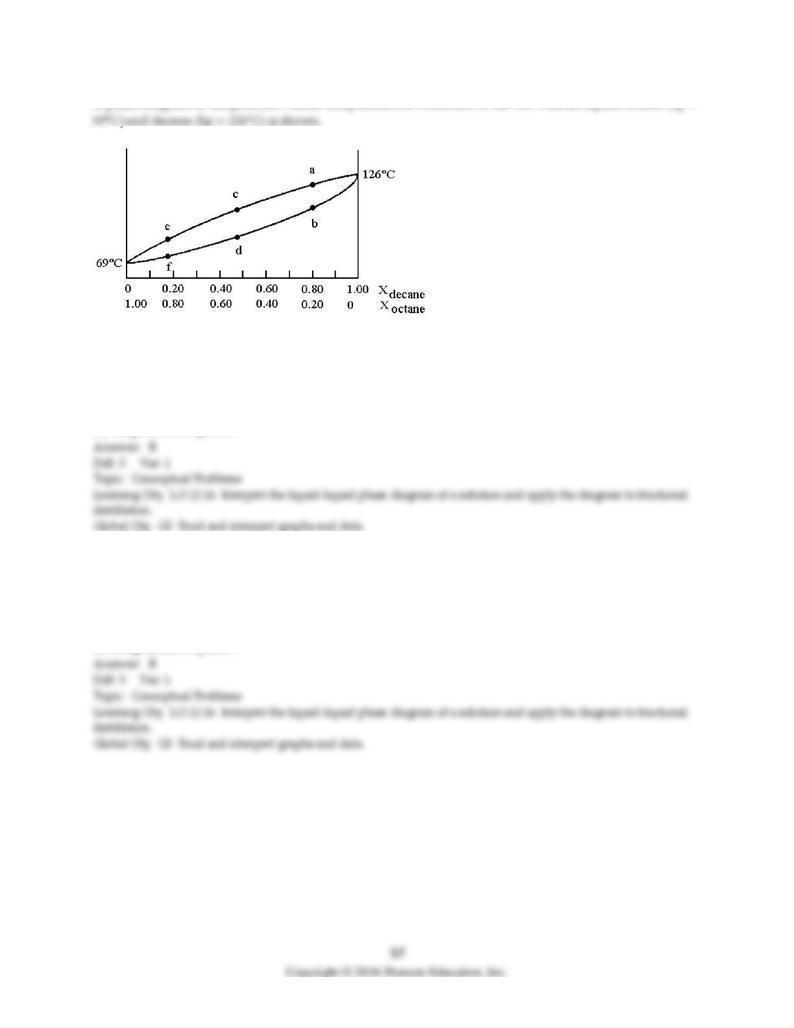
A phase diagram of temperature versus composition for a mixture of the two volatile liquids octane (bp =
105) Assume that you start with a mixture containing 0.80 mol of decane and 0.20 mol of octane, at what
approximate temperature will the mixture begin to boil?
A) temperature at point a
B) temperature at point b
C) temperature at point d
D) temperature at point f
106) Assume that you start with a mixture containing 0.80 mol of decane and 0.20 mol of octane, what is
the liquid composition at the boiling point?
A) 100% decane
B) composition at point b
C) composition at point c
D) composition at point e

107) Assume that you start with a mixture containing 0.80 mol of decane and 0.20 mol of octane, what is
the vapor composition at the boiling point?
A) 100% decane
B) composition at point b
C) composition at point c
D) composition at point e
108) Assume that the vapor at point c is condensed and reboiled. What is the liquid composition of the
condensed vapor prior to reboiling?
A) 100% decane
B) composition at point b
C) composition at point d
D) composition at point e
109) Assume that the vapor at point c is condensed and reboiled. What is the vapor composition during
reboiling?
A) 100% decane
B) composition at point b
C) composition at point c
D) composition at point e
110) Assume that the vapor at point c is condensed and reboiled. What is the boiling point?
A) temperature at point b
B) temperature at point c
C) temperature at point d
D) temperature at point f

111) What is the approximate boiling temperature of a mixture that is 0.70 XA and 0.30 XB?
A) 50°C
B) 75°C
C) 95°C
D) 100°C
112) What is the approximate vapor composition above a boiling solution that is 0.70 XA and 0.30 XB?
A) 0.70 XA, 0.30 XB
B) 0.50 XA, 0.50 XB
C) 0.30 XA, 0.70 XB
D) 0 XA, 1.00 XB

113) What is the approximate boiling temperature of the liquid formed from the condensation of the
vapor above a boiling solution that is 0.70 XA and 0.30 XB?
A) 50°C
B) 55°C
C) 90°C
D) 100°C
114) At 80°C, pure liquid A has a vapor pressure of 700 mm Hg and pure liquid B has a vapor pressure of
940 mm Hg. What is XA for a solution of A and B with a normal boiling point of
A) 0.25
B) 0.50
C) 0.75
D) A solution of A and B cannot boil at 80°C.
1) A gold ring is an example of a ________ solution.
A) gas/gas
B) liquid/solid
C) solid/lliquid
D) solid/solid
2) Which is not a solution?
A) sterling silver
B) fog
C) hydrochloric acid
D) coffee

3) KI does not dissolve well in nonpolar solvents because
A) solute-solute interactions are much larger than solvent-solvent or solute-solvent interactions.
B) solvent-solvent interactions are much larger than solute-solvent or solute-solute interactions.
C) solute-solvent interactions are much larger than solvent-solvent or solute-solute interactions.
D) solute-solvent interactions are similar to solvent-solvent and solute-solute interactions.
4) In the process of dissolving ionic compounds, the cations and anions are separated from the crystal
lattice and surrounded by an ordered shell of solvent molecules. If the solvent is water, the dissolved ions
are said to be
A) ionized.
B) homogenized.
C) hybridized.
D) hydrated.
5) Which cation in each set is expected to have the larger (more negative) hydration energy?
I. Li+ or Na+
II. Rb+ or Cu2+
A) Li+ in set I and Rb+ in set II
B) Li+ in set I and Cu2+ in set II
C) Na+ in set I and Rb+ in set II
D) Na+ in set I and Cu2+ in set II

6) Which cation in each set would be expected to have the larger (more negative) hydration energy?
I. Cu+or Cu2+
II. Li+ or NH4+
A) Cu+ in set I and Li+ in set II
B) Cu+ in set I and NH4+ in set II
C) Cu2+ in set I and Li+ in set II
D) Cu2+ in set I and NH4+ in set II
7) Arrange the following compounds in order of their expected increasing solubility in water:
NaF, CH3CH2-O-CH2CH3, CH3CH2CH2CH2-OH, CH3CH2CH2CH3.
A) CH3CH2CH2CH3 < NaF < CH3CH2-O-CH2CH3 < CH3CH2CH2CH2-OH
B) NaCl < CH3CH2-O-CH2CH3 < CH3CH2CH2CH2-OH < CH3CH2CH2CH3
C) CH3CH2CH2CH3 < CH3CH2-O-CH2CH3 < NaF < CH3CH2CH2CH2-OH
D) CH3CH2CH2CH3 < CH3CH2-O-CH2CH3 < CH3CH2CH2CH2-OH < NaF
8) What are the major solute-solvent interactions created when LiF dissolves in water?
A) dipole-ion
B) dispersion
C) hydrogen bonding
D) ion-dipole
9) What are the major solute-solvent interactions created when HOCH2CH2OH dissolves in water?
A) dipole-dipole
B) dispersion
C) hydrogen bonding
D) ion-dipole

10) Substances with high lattice energies tend to be less soluble than substances with low lattice energies.
On that basis predict the relative aqueous solubility at 20°C, from highest to lowest, of the following ionic
compounds:
A) Fe2(SO4)3 > Li2SO4 > KBr > LiCl
B) Fe2(SO4)3 > Li2SO4 > LiCl > KBr
C) KBr > LiCl > Li2SO4 > Fe2(SO4)3
D) LiCl > KBr > Li2SO4 > Fe2(SO4)3
11) What is the mole fraction of ethanol in a solution made by dissolving 7.30 g of ethanol, C2H5OH, in
53.6 g of water?
A) 0.0505
B) 0.0531
C) 0.120
D) 0.136
12) What is the mole fraction of I2 in a solution made by dissolving 139 g of I2 in 245 g of hexane, C6H14?
A) 0.161
B) 0.193
C) 0.278
D) 0.385
13) What is the mole fraction of oxygen in a gas mixture that is 27% oxygen and 73% nitrogen by volume?
A) 0.24
B) 0.27
C) 0.32
D) 0.37

14) What is the weight percent of vitamin C in a solution made by dissolving 1.30 g of vitamin C,
C6H8O6, in 55.0 g of water?
A) 0.195%
B) 0.242%
C) 2.31%
D) 2.36%
15) What is the weight percent of a caffeine solution made by dissolving 4.35 g of caffeine, C8H10N4O2,
in 75 g of benzene, C6H6?
A) 0.055%
B) 0.058%
C) 5.5%
D) 5.8%
16) What volume of a 0.716 M KBr solution is needed to provide 10.5 g of KBr?
A) 7.52 mL
B) 14.7 mL
C) 63.2 mL
D) 123 mL
17) What volume of 3.00 M CH3OH solution is needed to provide 0.320 mol of CH3OH?
A) 1.04 mL
B) 9.38 mL
C) 107 mL
D) 960 mL

18) How much water must be added to 36.0 g of CaCl2 to produce a solution that is 35.0 wt% CaCl2?
A) 48.6 g
B) 66.9 g
C) 97.2 g
D) 103 g
19) To make a 0.125 M solution, one could take 0.125 moles of solute and add
A) 1.00 L of solvent.
B) 1.00 kg of solvent.
C) enough solvent to make 1.00 L of solution.
D) enough solvent to make 1.00 kg of solution.
20) To make a 0.125 m solution, one could take 0.125 moles of solute and add
A) 1.00 L of solvent.
B) 1.00 kg of solvent.
C) enough solvent to make 1.00 L of solution.
D) enough solvent to make 1.00 kg of solution.
21) What molality of pentane is obtained by dissolving 25 g pentane, C5H12, in 245.0 g hexane, C6H14?
A) 0.093 m
B) 0.11 m
C) 1.4 m
D) 100 m

22) A solution is 2.25% by weight NaHCO3. How many grams of NaHCO3 are in 250.0 g of solution?
A) 0.900 g
B) 5.62 g
C) 117 g
D) 225 g
23) How many grams of KBr are required to make 250. mL of a 0.115 M KBr solution?
A) 0.242 g
B) 2.17 g
C) 3.42 g
D) 28.8 g
24) What is the molality of a glucose solution prepared by dissolving 27.0 g of glucose, C6H12O6, in 125.9
g of water?
A) 1.19 × 10-3 m
B) 0.214 m
C) 0.981 m
D) 1.19 m
25) A solution of LiCl in water has XLiCl = 0.0800. What is the molality?
A) 4.01 m LiCl
B) 4.44 m LiCl
C) 4.83 m LiCl
D) 8.70 m LiCl

26) A solution of LiCl in water is 22.0 wt% LiCl. What is the mole fraction of LiCl?
A) 0.107
B) 0.120
C) 0.519
D) 4.33
27) A solution has a density of 1.023 g/mL and a concentration of 0.0800 g/dL. What is the concentration
in parts per million?
A) 700 ppm
B) 1408 ppm
C) 550 ppm
D) 818 ppm
28) Fresh air contains approximately 400 ppm CO2, whereas the breath of an intoxicated person contains
about 4 percent CO2. The amount of CO2 in the breath of an intoxicated person is ________ times the
amount of CO2 in fresh air.
A) 10-2
B) 10-1
C) 101
D) 102

29) The dose of amoxicillin given to a young child is 40.0 mg/kg of body weight/day. If the amoxicillin is
administered as a suspension having a concentration of 400. mg/5 mL, how many mL of amoxicillin must
be administered per dose for a child weighing 30.0 pounds?
A) 3.00 mL
B) 6.82 mL
C) 16.0 mL
D) 9.9 mL
30) Molarity is defined as moles of solute per
A) kilogram of solvent.
B) liter of solution.
C) mole of solute.
D) total moles present.
31) Molality is defined as moles of solute per
A) kilogram of solvent.
B) liter of solvent.
C) mole of solvent.
D) total moles present.
32) Which concentration becomes smaller as the temperature is increased from 25°C to 60°C?
A) mass %
B) molality
C) molarity
D) mole fraction

33) What is the molality of ethanol in a solution made by dissolving 7.30 g of ethanol, C2H5OH, in 53.6 g
of water?
A) 0.00550 m
B) 0.500 m
C) 2.96 m
D) 400 m
34) A 1.40 M solution of CaCl2 in water has a density of 1.11 g/mL. What is the molality?
A) 1.90 m CaCl2
B) 2.30 m CaCl2
C) 1.47 m CaCl2
D) 1.44 m CaCl2
35) Calculate the molality of a solution with 53 grams of KCl in 2000 mL of water. (density of water is 1.00
g/mL).
A) 0.027
B) 0.27
C) 0.00071
D) 0.0027
36) In which case should CO2(g) be more soluble in water?
A) The total pressure is 5 atm and the partial pressure of CO2 is 0.4 atm.
B) The total pressure is 3 atm and the partial pressure of CO2 is 0.6 atm.
C) The total pressure is 1 atm and the partial pressure of CO2 is 0.3 atm.
D) The total pressure is 1 atm and the partial pressure of CO2 is 0.5 atm.

37) The Henry's Law constant of methyl bromide, CH3Br, is k = 0.159 mol/(L ∙ atm) at 25°C. What is the
solubility of methyl bromide in water at 25°C and at a partial pressure of 290. mm Hg?
A) 0.0607 mol/L
B) 0.382 mol/L
C) 0.417 mol/L
D) 46.1 mol/L
38) A saturated solution is defined as
A) a concentrated solution.
B) a solution that is in equilibrium with pure solute.
C) a solution than is in equilibrium with undissolved solute.
D) a solution that is not in equilibrium with both pure solvent and undissolved solute.
39) Which of the following does not affect the solubility of a solute in a given solvent?
A) hydrogen bonding of the solute
B) nonpolarity of the solvent
C) rate of stirring
D) temperature of the solvent and solute
40) In which case should CO2(g) be more soluble in water?
A) The total pressure is 6 atm and the partial pressure of CO2 is 1 atm.
B) The total pressure is 4 atm and the partial pressure of CO2 is 3 atm.
C) The total pressure is 1 atm and the partial pressure of CO2 is 0.03 atm.
D) The total pressure is 1 atm and the partial pressure of CO2 is 0.4 atm.

41) A solution is prepared by dissolving 50.0 g of sucrose, C12H22O11, in 250. g of water at 25°C. What is
the vapor pressure of the solution if the vapor pressure of water at 25°C is 23.76 mm Hg?
A) 0.247 mm Hg
B) 19.8 mm Hg
C) 23.5 mm Hg
D) 24.0 mm Hg
42) A KCl solution is prepared by dissolving 25.0 g KCl in 250.0 g of water at 25°C. What is the vapor
pressure of the solution if the vapor pressure of water at 25°C is 23.76 mm Hg?
A) 21.6 mm Hg
B) 22.7 mm Hg
C) 23.2 mm Hg
D) 24.9 mm Hg
43) At a given temperature the vapor pressures of benzene and toluene are 183 mm Hg and 59.2 mm Hg,
respectively. Calculate the total vapor pressure over a solution of benzene and toluene with Xbenzene =
0.520.
A) 95.2 mm Hg
B) 121 mm Hg
C) 124 mm Hg
D) 242 mm Hg
44) If one mole of each is dissolved in 1.00 L of water, which will lower the vapor pressure the most?
A) C12H22O11
B) KNO3
C) C3H7OH
D) MgCl2

45) Which of the following solutions will have the lowest freezing point?
A) 0.010 m Rb I
B) 0.010 m K2SeO4
C) 0.035 m CH3OH
D) 0.015 m SrBr2
46) What is the expected freezing point of a 0.50 m solution of K2SO4 in water? Kf for water is 1.86°C/m.
A) -0.93°C
B) -1.9°C
C) -2.8°C
D) -6.5°C
47) Calculate the freezing point of a solution of 30.0 g methyl salicylate, C7H6O2, dissolved in 800. g of
benzene, C6H6. Kf for benzene is 5.10°C/m and the freezing point is 5.50°C for benzene.
A) -1.56°C
B) 1.56°C
C) 3.93°C
D) 7.06°C
48) What is the freezing point of a solution of 2.86 g MgCl2 in 100 g of water? Kf for water is 1.86°C/m.
A) -0.0558°C
B) -0.558°C
C) -1.12°C
D) -1.67°C

49) An aqueous solution has a normal boiling point of 102.5°C. What is the freezing point of this solution?
For water Kb = 0.51°C/m and Kf = 1.86°C/m.
A) -0.69°C
B) -2.5°C
C) -3.6°C
D) -9.1°C
50) When 2.36 g of a nonvolatile solute is dissolved in 100 g of solvent, the largest change in freezing
point will be achieved when the solvent is
A) paradichlorobenzene, Kf = 7.10.
B) tert-butanol, Kf = 9.1.
C) water, Kf = 1.86.
D) All are expected to have the same freezing point.
51) A 0.50 m solution of which solute has the largest van't Hoff factor?
A) CaF2
B) Li3PO4
C) K2CO3
D) NaNO3
52) A 0.020 m aqueous solution containing which solute will have the lowest freezing point?
A) LiCl
B) NaF
C) KCl
D) All will have approximately the same freezing point.

53) A 0.35 m aqueous solution of an unknown solute has a boiling point elevation of 0.79°C. The boiling
point elevation of a 0.35 m solution of a nonionizing molecular solute in water is 0.33°C. How many
moles of particles are formed per mole of solute when the unknown solute is dissolved in water?
A) 1.4
B) 2.0
C) 2.4
D) 3.0
54) Chloroform has a boiling point of 61.1°C and dichloromethane has a boiling point of 40.0°C. When
0.250 mol of dichloromethane is added to 0.750 mol of chloroform, the resulting solution will have a
boiling point
A) between 40.0°C and 61.1°C, but closer to 61.1°C.
B) between 40.0°C and 61.1°C, but closer to 40.0°C.
C) greater than 61.1°C.
D) less than 40.0°C.
55) Which of the following pairs of solutions have roughly the same boiling point elevation?
A) 0.100 m C6H12O6 and 0.0333 m CuF2
B) 0.100 m KF and 0.100 m C6H12O6
C) 0.200 m KCl and 0.300 m Li2SO4
D) 0.100 m LiBr and 0.0500 m MgBr2
56) Calculate the freezing point of a solution when 370 grams of Ca3(PO4) is added to 3000mL of water.
(density of water is 1.00 g/mL). The Kf for water is 1.86°C/m and normal freezing point of water is 0.00°C.
A) -0.74
B) -0.0030
C) -3.70
D) -1.27

57) A solution is made by dissolving 19.5 g of sucrose, C12H22O11, in 117 g of water, producing a
solution with a volume of 125 mL at 20°C. What is the expected osmotic pressure at 20°C?
A) 11.0 atm
B) 23.2 atm
C) 50.0 atm
D) 58 atm
12.3 Short Answer Questions
1) In a solution that is 75% ethyl alcohol and 25% water, the solute is ________ and the solvent is
________.
2) According to the rule of thumb "like dissolves like" ionic solids like LiF or polar substances like glucose
are more soluble in ________ solvents, whereas nonpolar substances like I2 are more soluble in ________
solvents.
3) When KI is dissolved in water, the major forces overcome are ________ forces in the solute and
________ forces in the solvent, and the major forces created between solute and solvent particles are
________ forces.
4) A person is considered legally intoxicated with a blood alcohol level of 80 mg/dL. Assuming that blood
plasma has a density of 1.025 g/mL, what is this concentration expressed in parts per million?

5) Amphetamines in urine can be confirmed by mass spectroscopy at a concentration of 500 ng/mL.
Assuming a urine density of 1.025 g/mL, what is this concentration in parts per million?
6) Molarity is defined as ________, whereas molality is defined as ________.
7) The number of moles of ions in 35.0mL of 4.00 M Na2SO4 is ________ moles.
8) The volume of 0.200 M H2SO4 that contains 5.00 mmol of H+ is ________ mL.
9) Cocaine, C17H21NO4, in urine can be confirmed by mass spectroscopy at a concentration of 150
ng/mL. Assuming a urine density of 1.025 g/mL, what is this concentration expressed as molality?
10) Concentrations of fluoride in drinking water greater than 4.0 mg/L can cause mottled teeth in
children. What is 4.0 mg/L expressed as molality?
11) The solubility of a gas in a liquid is greatest at ________ pressures and ________ temperatures.

12) Dinitrogen monoxide is still used as a general anesthetic. The Henry's law constant for the solubility
of N2O in water is 2.5 × 10-2 mol/L·atm. What is the molar solubility of N2O in water if the partial
pressure of N2O is 1700 mm Hg?
13) At 20°C and 0.28 atm pressure Xenon has a solubility in water of 1.4 mmol/L. What is the Henry's
Law-constant in mol/ L·atm at 20°C?
14) Freezing point depression, boiling point elevation, vapor pressure lowering, and osmotic pressure are
examples of ________ properties, which depend on the amount but not the chemical identity of dissolved
particles.
15) When solute is added to water, the solution has a ________ freezing point than that of pure ice at 0˚C.
16) ________ properties depend only on the identity of the solute and on the number of solute particles
present.
17) At 80.0°C heptane, C7H16, has a vapor pressure of 428 mm Hg and octane, C8H18, has a vapor
pressure of 175 mm Hg. What is the vapor pressure of a solution that contains 20.0 g C7H16 and 11.4 g
C8H18?

18) At 80.0°C benzene has a vapor pressure of 96.0 mm Hg and toluene has a vapor pressure of 30.3 mm
Hg. If a mixture of benzene and toluene has a vapor pressure of 54.6 mm Hg, what are the mole fractions
of benzene and toluene?
19) If dissociation of MgCl2 in water were 100%, the van't Hoff factor would be ________; however, for
real solutions the van't Hoff factor for MgCl2 is ________ (greater than, less than) this value.
20) The vapor pressure of water at 25°C is 23.8 mm Hg. The vapor pressure lowering, ΔP, of an aqueous
solution containing 0.500 mole fraction NaCl is ________ (equal to, greater than, less than) 11.9 mm Hg?
21) At 25°C the vapor pressures of benzene and toluene are 96.0 mm Hg and 30.5 mm Hg, respectively.
The vapor pressure of a solution that has a 0.50 mole fraction of toluene will have a vapor pressure
________ (equal to, greater than, less than) 30.5 mm Hg.
22) The boiling point of a solution containing a nonvolatile solute will be ________ than the boiling point
of the pure solvent, whereas the boiling point of a solution of two volatile liquids will be ________ than
the boiling point of the more volatile liquid and ________ than the boiling point of the less volatile liquid.
23) A 1.0 m aqueous BaI2 solution will have a ________ vapor pressure, ________ freezing point, and
________ boiling point than a 1.0 m aqueous solution of LiI.

24) The osmotic pressure of a 0.10 M solution of sucrose, C12H22O11 will be ________ (equal to, greater
than, less than) the osmotic pressure of a 0.10 M solution of fructose, C6H12O6.
25) The osmotic pressure of a 100-mL solution containing 1.0 g of sucrose, C12H22O11 will be ________
(equal to, greater than, less than) the osmotic pressure of a 100-mL solution containing 1.0 g of fructose,
C6H12O6.
26) At 300 K a 500.0 mL solution containing 0.4314 g of dextrose has an osmotic pressure of 89.6 mm Hg.
What is the molar mass of dextrose?
27) At 25°C the vapor pressures of benzene and toluene are 96.0 mm Hg and 30.5 mm Hg, respectively.
When a 1:1 molar mixture of benzene and toluene is fractionally distilled, the first fraction will have a
mole fraction of benzene that is ________ (equal to, greater than, less than) 0.50.