
Chemistry, 7e (McMurry/Fay)
Chapter 15 Aqueous Equilibria: Acids and Bases
15.1 Multiple-Choice Questions
1) When dissolved in water, which of the following compounds is an Arrhenius acid?
A) HCN
B) NaOH
C) NaF
D) CH3CH2OH
2) What are the Br∅nsted-Lowry bases in the following chemical reaction?
C5H5N(aq) + H2O(l) ⇌ C5H5NH+(aq) + OH-(aq)
A) C5H5N, H2O
B) C5H5N, C5H5NH+
C) C5H5N, OH-
D) C5H5N, H2O, OH-
3) What are the Br∅nsted-Lowry acids in the following chemical reaction?
HBr(sol) + CH3COOH(sol) ⇌ CH3C(OH)2+(sol) + Br-(sol)
A) HBr, CH3COOH
B) HBr, CH3C(OH)2+
C) CH3COOH, CH3C(OH)2+
D) CH3COOH, Br-

4) What are the conjugate acid-base pairs in the following chemical reaction?
NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq)
A) NH3, H2O and NH4+, OH-
B) NH3, NH4+ and H2O, OH-
C) NH3, OH- and H2O, NH4+
D) NH3 and NH4+
5) Identify the conjugate acid/base pairs present in an aqueous solution of hydrogen sulfate ion, HSO4-.
A) HSO4-/SO42- and H3O+/H2O
B) H2SO4/HSO4- and H2O/OH-
C) HSO4-/H2O and H3O+/SO42-
D) HSO4-/H2O and H2SO4/OH-
6) What is the conjugate base of the Br∅nsted-Lowry acid HPO42-?
A) H3PO4
B) H2PO4-
C) HPO42-
D) PO43-

7) What is the conjugate acid of the Br∅nsted-Lowry base HAsO42-?
A) H2AsO4-
B) AsO43-
C) H2O
D) H3O+
8) Write a balanced equation for the dissociation of the Br∅nsted-Lowry acid HSO4- in water.
A) HSO4-(aq) + H2O(l) ⇌ H2SO4(aq) + OH-(aq)
B) HSO4-(aq) + H2O(l) ⇌ SO42-(aq) + H3O+(aq)
C) HSO4-(aq) + H2O(l) ⇌ SO32-(aq) + OH-(aq)
D) HSO4-(aq) + H2O(l) ⇌ SO3(g) + OH-(aq) + H2O(l)
9) Which of the following Br∅nsted-Lowry acids does not behave as a strong acid when it is dissolved in
water?
A) HBr
B) HCl
C) HNO2
D) HClO4

10) What is the strongest Br∅nsted-Lowry acid in the chemical reaction shown below?
2 HNO3(aq) + Ba(OH)2(aq) → Ba(NO3)2(aq) + 2 H2O(l)
A) HNO3
B) Ba(OH)2
C) Ba(NO3)2
D) H2O
11) From the following chemical reactions determine the relative Br∅nsted-Lowry acid strengths
(strongest to weakest).
HCl(aq) + H2O(l) → H3O+(aq) + Cl-(aq)
HCN(aq) + H2O(l) ⇌ H3O+(aq) + CN-(aq)
A) HCl > HCN > H3O+
B) HCl > H3O+ > HCN
C) H3O+ > HCl > HCN
D) HCN > H3O+ > HCl

12) From the following chemical reactions determine the relative Br∅nsted-Lowry acid strengths
(strongest to weakest).
HClO4(aq) + H2O(l) → H3O+(aq) + ClO4-(aq)
HNO2(aq) + H2O(l) ⇌ H3O+(aq) + NO2-(aq)
A) HClO4 > H3O+ > HNO2
B) HClO4 > HNO2 > H3O+
C) H3O+ > HClO4 > HNO2
D) H3O+ > HNO2 > HClO4
13) From the following chemical reactions determine the relative Br∅nsted-Lowry acid strengths
(strongest to weakest).
HClO4(sol) + CH3COOH(l) → CH3C2(OH)2+(sol) + ClO4-(aq)
H2SO4(sol) + CH3COOH(l) ⇌ CH3C(OH)2+(sol) + HSO4-(sol)
A) HClO4 > H2SO4 > CH3COOH
B) HClO4 > H2SO4 > CH3C(OH)2+
C) HClO4 > CH3COOH > H2SO4
D) HClO4 > CH3C(OH)2+ > H2SO4

14) From the following chemical reactions determine the relative Br∅nsted-Lowry base strengths
(strongest to weakest).
HNO3(aq) + H2O(l) → H3O+(aq) + NO3-(aq)
HF(aq) + H2O(l) ⇌ H3O+(aq) + F-(aq)
A) HNO3 > H3O+ > HF
B) NO3- > H2O > F-
C) F- > H2O > NO3-
D) F- > NO3- > H2O
15) From the following chemical reactions determine the relative Br∅nsted-Lowry base strengths
(strongest to weakest).
2 NaH(s) + H2O(l) → 2 NaOH + H2(g)
NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq)
A) H- > OH- > NH3
B) H- > NH3 > OH-
C) OH-> H- > NH3
D) OH- > NH3 > H-

16) The equilibrium constant, K, for the reaction shown below has a value 1.8 × 10-5. In this reaction
which is the strongest acid and which is the strongest base?
CH3CO2H(aq) + H2O(l) ⇌ H3O+(aq) + CH3CO2-(aq)
A) CH3CO2H and CH3CO2-
B) CH3CO2H and H2O
C) H3O+ and H2O
D) H3O+ and CH3CO2-
17) Determine the strongest acid of the set.
A) A
B) B
C) C
D) D

18) Arrange the acids in order of increasing acid strength.
A) D < C < A < B
B) D < C < B < A
C) D < B < A < C
D) D < A < C < B
19) Using the conjugate acid-base pairs listed below, complete the following equation with the pair that
gives an equilibrium constant Kc > 1.
________ + HSO3- ⇌ ________ + H2SO3
A) CH3CO2H/ CH3CO2-
B) HCO2H/ HCO2-
C) HNO2/NO2-
D) HNO3/NO3-
20) Using the conjugate acid-base pairs listed below, complete the following equation with the pair that
gives an equilibrium constant Kc > 1.
________ + H2CO3 ⇌ ________ + HCO3-
A) HF/F-
B) HCl/Cl-
C) HOCl/OCl-
D) HSO42-/SO42-

21) What is the strongest acid among the following?
A) HF
B) HCl
C) HBr
D) HI
22) What is the strongest acid among the following?
A) H2O
B) H2S
C) H2Se
D) H2Te
23) What is the weakest acid among the following?
A) SiH4
B) PH3
C) H2S
D) HCl
24) What is the strongest acid of the following?
A) HOI
B) HOBr
C) HOCl
D) All are equivalent.

25) Which of the following should have the lowest bond strength?
A) HOI
B) HOCl
C) HOAt
D) HOBr
26) An acidic solution at 25°C has
A) [H3O+] > [OH-] > 1 × 10-7 M.
B) [H3O+] > 1 × 10-7 M > [OH-].
C) [H3O+] = [OH-] > 1 × 10-7 M.
D) [H3O+] < 1 × 10-7 M > [OH-].
27) At 50°C the value of Kw is 5.5 × 10-14. A basic solution at 50°C has
A) [H3O+] < [OH-] < 2 × 10-7 M.
B) [H3O+] < 2 × 10-7 M < [OH-].
C) [H3O+] = [OH-] < 2 × 10-7 M.
D) [H3O+] > 2 × 10-7 M < [OH-].
28) Calculate the hydronium ion concentration in an aqueous solution that contains 2.50 × 10-4 M in
hydroxide ion.
A) 4.00 × 10-9 M
B) 4.00 × 10-10 M
C) 4.00 × 10-11 M
D) 5.00 × 10-11 M

29) Calculate the hydroxide ion concentration in an aqueous solution that contains 3.50 × 10-3 M in
hydronium ion.
A) 2.86 × 10-4 M
B) 2.86 × 10-11 M
C) 2.86 × 10-12 M
D) 3.50 × 10-12 M
30) A solution with a hydrogen ion concentration of 3.25 × 10-2 M is ________ and has a hydroxide
concentration of ________.
A) acidic, 3.08 × 10-12 M
B) acidic, 3.08 × 10-13 M
C) basic, 3.08 × 10-12 M
D) basic, 3.08 × 10-13 M
31) A solution with a hydroxide ion concentration of 4.15 × 10-4 M is ________ and has a hydrogen ion
concentration of ________.
A) acidic, 2.41 × 10-10 M
B) acidic, 2.41 × 10-11 M
C) basic, 2.41 × 10-10 M
D) basic, 2.41 × 10-11 M

32) If the ionization constant of water, Kw, at 40°C is 2.92 × 10-14, then what is the hydronium ion
concentration for a neutral solution?
A) [H3O+] < 1.00 × 10-7 M
B) [H3O+] > 1.71 × 10-7 M
C) [H3O+] = 1.71 × 10-7 M
D) [H3O+] < 1.71 × 10-7 M
33) Human tears have a concentration of H3O+ that is 3.16 × 10-8. The concentration of OH- in human
tears is
A) greater than 3.16 × 10-7 and tears are acidic.
B) greater than 3.16 ×10-7 and tears are basic.
C) less than 3.16 × 10-7 and tears are acidic.
D) less than 3.16 × 10-7 and tears are basic.
34) An acidic solution at 25°C will have a hydronium ion concentration ________ and a pH value
________.
A) [H3O+] > 1 × 10-7 M, pH > 7.00
B) [H3O+] > 1 × 10-7 M, pH < 7.00
C) [H3O+] < 1 × 10-7 M, pH > 7.00
D) [H3O+] < 1 × 10-7 M, pH < 7.00

35) If the ionization constant of water, Kw, at 40°C is 2.92 × 10-14, then what is the hydronium ion
concentration and pH for an acidic solution?
A) [H3O+] > 1.71 × 10-7 M and pH > 6.77
B) [H3O+] > 1.71 × 10-7 M and pH < 6.77
C) [H3O+] < 1.71 × 10-7 M and pH > 6.77
D) [H3O+] < 1.71 × 10-7 M and pH < 6.77
36) Calculate the pH for an aqueous solution of acetic acid that contains 2.15 × 10-3 M hydronium ion.
A) 4.65 × 10-12
B) 2.15 × 10-3
C) 2.67
D) 11.33
37) Calculate the pH for an aqueous solution of pyridine that contains 2.15 × 10-4 M hydroxide ion.
A) 4.65 × 10-11
B) 2.15 × 10-4
C) 3.67
D) 10.33
38) What is the hydroxide ion concentration and the pH for a hydrochloric acid solution that has a
hydronium ion concentration of 1.50 × 10-4 M?
A) 6.67 × 10-10 M, 4.82
B) 6.67 × 10-10 M, 9.18
C) 6.67 × 10-11 M, 3.82
D) 6.67 × 10-11 M, 10.18

39) What is the hydronium ion concentration and the pH for an aqueous solution of NH3 that has a
hydroxide ion concentration of 2.25 × 10-3 M?
A) 4.44 × 10-11 M, 3.65
B) 4.44 × 10-11 M, 10.35
C) 4.44 × 10-12 M, 2.65
D) 4.44 × 10-12 M, 11.35
40) What is the hydronium ion concentration of an acid rain sample that has a pH of 3.15?
A) 1.41 × 10-11 M
B) 7.08 × 10-4 M
C) 3.15 M
D) 10.85 M
41) What is the hydroxide ion concentration of a lye solution that has a pH of 11.20?
A) 6.31 × 10-12 M
B) 1.58 × 10-3 M
C) 2.80 M
D) 11.20 M
42) At 25°C, the pH of a vinegar solution is 2.60. What are the values of [H3O+] and [OH-] in the solution?
A) 3.99 × 10-12 M, 2.51 × 10-3 M
B) 2.51 × 10-3 M, 3.98 × 10-12 M
C) 2.51 × 10-3 M, 11.40 M
D) 2.60 M, 11.40 M

43) Which of the following solutions has the highest concentration of hydroxide ions [OH-]?
A) pH = 3.21
B) pH = 7.83
C) pH = 10.93
D) pH = 12.04
44) Which of the following indicates the most basic solution?
A) [H+] = 1 × 10-10 M
B) pOH = 6.7
C) [OH-] = 7 × 10-5 M
D) pH = 4.2
45) What is the approximate pH of a solution X that gives the following responses with the indicators
shown?
Indicators
HIn — In-
pH range
Solution X
methyl orange
red-yellow
3.2-4.4
yellow
methyl red
red-yellow
4.8-6.0
yellow
bromothymol blue
yellow-blue
6.0-7.6
green
phenolphthalein
colorless-pink
8.2-10.0
colorless
A) 3.2 - 4.4
B) 4.8 - 6.0
C) 6.0 - 7.6
D) 8.2 - 10.0

46) What is the approximate pH of a solution X that gives the following responses with the indicators
shown?
Indicators
HIn — In-
pH range
Solution X
methyl orange
red-yellow
3.2-4.4
yellow
methyl red
red-yellow
4.8-6.0
orange
bromothymol blue
yellow-blue
6.0-7.6
yellow
phenolphthalein
colorless-pink
8.2-10.0
colorless
A) 3.2 - 4.4
B) 4.8 - 6.0
C) 6.0 - 7.6
D) 8.2 - 10.0
47) What is the approximate pH of a solution X that gives the following responses with the indicators
shown?
Indicators
HIn — In-
pH range
Solution X
methyl orange
red-yellow
3.2-4.4
yellow
methyl red
red-yellow
4.8-6.0
yellow
bromothymol blue
yellow-blue
6.0-7.6
blue
phenolphthalein
colorless-pink
8.2-10.0
pink
A) 4.8 - 6.0
B) 6.0 - 7.6
C) 7.6 - 8.2
D) > 8.2

48) What is the pH of a 0.020 M HClO4 solution?
A) 0.020
B) 0.040
C) 1.70
D) 12.30
49) What is the pH of a 0.020 M Ba(OH)2 solution?
A) 1.40
B) 1.70
C) 12.30
D) 12.60
50) What is the pH of a solution prepared by dissolving 0.15 gram of solid CaO (lime) in enough water to
make 2.00 L of aqueous Ca(OH)2 (limewater)?
CaO(s) + H2O(l) → Ca2+(aq) + 2 OH-(aq)
A) 2.57
B) 2.87
C) 11.13
D) 11.43
51) What is the pH of a solution prepared by diluting 25.00 mL of 0.10 M HCl with enough water to
produce a total volume of 100.00 mL?
A) 1.00
B) 1.60
C) 2.00
D) 3.20

52) What is the pH of a solution prepared by diluting 25.00 mL of 0.020 M Ba(OH)2 with enough water to
produce a total volume of 250.00 mL?
A) 2.40
B) 2.70
C) 11.30
D) 11.60
53) What is the pH of a solution made by mixing 100.00 mL of 0.20 M HCl with 50.00 mL of 0.10 M HCl?
Assume that the volumes are additive.
A) 0.15
B) 0.52
C) 0.78
D) 1.70
54) What is the pH of a solution made by mixing 100.0 mL of 0.10 M HNO3, 50.0 mL of 0.20 M HCl, and
100.0 mL of water? Assume that the volumes are additive.
A) 0.30
B) 0.82
C) 1.00
D) 1.10

55) What is the equilibrium constant expression (Ka) for the acid dissociation of hydrocyanic acid HCN?
The equation of interest is
HCN(aq) + H2O(l) ⇌ H3O+(aq) + CN-(aq).
A) Ka = ([H3O+][CN-])/([HCN][H2O])
B) Ka = ([H3O+][CN-])/([HCN])
C) Ka = ([HCN][H2O])/([H3O+][CN-])
D) Ka = ([HCN])/([H3O+][CN-])
56) What is the equilibrium constant expression (Ka) for the acid dissociation of nitrous acid HNO2? The
equation of interest is
HNO2(aq) + H2O(l) ⇌ H3O+(aq) + NO2-(aq).
A) Ka = ([H3O+][NO2-])/([HNO2][H2O])
B) Ka = ([H3O+][NO2-])/([HNO2])
C) Ka = ([HNO2][H2O])/([H3O+][NO2-])
D) Ka = ([HNO2])/([H3O+][NO2-])

57) Determine the acid dissociation constant for a 0.10 M acetic acid solution that has a pH of 2.87. Acetic
acid is a weak monoprotic acid and the equilibrium equation of interest is
CH3COOH(aq) + H2O(l) ⇌ H3O+(aq) + CH3CO2-(aq).
A) 1.3 × 10-2
B) 1.3 × 10-3
C) 1.8 × 10-5
D) 1.8 × 10-6
58) Determine the acid dissociation constant for a 0.010 M nitrous acid solution that has a pH of 2.70.
Nitrous acid is a weak monoprotic acid and the equilibrium equation of interest is
HNO2(aq) + H2O(l) ⇌ H3O+(aq) + NO2-(aq).
A) 8.0 × 10-3
B) 2.0 × 10-3
C) 5.0 × 10-4
D) 4.0 × 10-4
59) Determine the acid dissociation constant for a 0.020 M formic acid solution that has a pH of 2.74.
Formic acid is a weak monoprotic acid and the equilibrium equation of interest is
HCOOH(aq) + H2O(l) ⇌ H3O+(aq) + HCO2-(aq).
A) 1.8 × 10-3
B) 1.8 × 10-4
C) 3.6 × 10-4
D) 3.6 × 10-5

60) The pH of 0.255 M HCN is 4.95. What is the value of Ka for hydrocyanic acid?
A) 1.3 × 10-10
B) 4.9 × 10-10
C) 1.1 × 10-5
D) 4.4 × 10-5
61) The pH of 0.150 M CH3CO2H, acetic acid, is 2.78. What is the value of Ka for acetic acid?
A) 2.8 × 10-6
B) 1.9 × 10-5
C) 1.7 × 10-3
D) 1.1 × 10-2
62) What is the strongest monoprotic acid of the following set if all the acids are at 0.100 M concentration?
A) hydrofluoric acid with Ka = 3.5 × 10-4
B) benzoic acid with Ka = 6.5 × 10-5
C) acetic acid with Ka = 1.8 × 10-5
D) hypochlorous acid with Ka = 3.5 × 10-8

63) What is the hydronium ion concentration of a 0.100 M acetic acid solution with a Ka = 1.8 × 10-5? The
equation for the dissociation of acetic acid is:
CH3CO2H(aq) + H2O(l) ⇌ H3O+(aq) + CH3CO2-(aq).
A) 1.3 × 10-2 M
B) 4.2 × 10-2 M
C) 1.3 × 10-3 M
D) 4.2 × 10-3 M
64) What is the hydronium ion concentration of a 0.100 M hypochlorous acid solution with Ka = 3.5 ×
10-8? The equation for the dissociation of hypochlorous acid is:
HOCl(aq) + H2O(l) ⇌ H3O+(aq) + OCl-(aq).
A) 1.9 × 10-4
B) 5.9 × 10-4
C) 1.9 × 10-5
D) 5.9 × 10-5
65) Para-Aminobenzoic acid (PABA), p-H2NC6H4(COOH), is used in some sunscreens and hair
conditioning products. Calculate the pH of an aqueous solution with [PABA] = 0.030 M and Ka = 2.2 ×
10-5.
A) 1.52
B) 3.09
C) 4.66
D) 6.18

66) A tablet containing 500.0 mg of aspirin (acetylsalicylic acid or HC9H7O4) was dissolved in enough
water to make 100 mL of solution. Given that Ka = 3.0 × 10-4 for aspirin, what is the pH of the solution?
A) 1.57
B) 2.54
C) 3.52
D) 5.08
67) Benzoic acid (C6H5CO2H = HBz) solutions are sometimes used in experiments to determine the
molarity of a basic solution of unknown concentration. What is the pH of a 0.100 M solution of benzoic
acid if Ka = 6.5 × 10-5 and the equilibrium equation of interest is HBz(aq) + H2O(l) ⇌ H3O+(aq) + Bz-(aq).
A) 1.00
B) 2.59
C) 4.19
D) 5.19
68) Potassium hydrogen phthalate (molar mass = 204.2 g/mol) is one of the most commonly used acids for
standardizing solutions containing bases. KHP is a monoprotic weak acid with Ka = 3.91 × 10-6. Calculate
the pH of the solution that results when 0.40 g of KHP is dissolved in enough water to produce 25.0 mL
of solution.
A) 2.10
B) 3.26
C) 4.30
D) 5.41

69) Vinegar is a 5.0% solution by weight of acetic acid (CH3CO2H) in water. Given that Ka = 1.8 × 10-5 for
acetic acid and assuming the density of vinegar to be 1.00 g/cm3, what is the pH of this vinegar solution?
A) 2.00
B) 2.41
C) 2.87
D) 4.74
70) Vinegar is a 5.0% solution by weight of acetic acid (CH3CO2H) in water. Given that the pH for acetic
acid is 2.41, the Ka = 1.8 × 10-5 and assuming the density of vinegar to be 1.00 g/cm3, what is the percent
dissociation of acetic acid in vinegar?
A) 0.47%
B) 1.5%
C) 4.0%
D) 5.0%
71) What is the percent dissociation of a benzoic acid solution with pH = 2.59? The acid dissociation
constant for this monoprotic acid is 6.5 × 10-5.
A) 0.50%
B) 1.5%
C) 2.5%
D) 3.5%

72) The percent dissociation of acetic acid changes as the concentration of the acid decreases. A 100-fold
decrease in acetic acid concentration results in a ________ fold ________ in the percent dissociation.
A) 10, increase
B) 10, decrease
C) 100, increase
D) 100, decrease
73) What is the second stepwise equilibrium constant expression for phosphoric acid H3PO4?
A) Ka2 = ([H3O+][H2PO4-])/([H3PO4])
B) Ka2 = ([H3O+]2[HPO42-])/([H3PO4])
C) Ka2 = ([H3O+]3[PO43-])/([H3PO4])
D) Ka2 = ([H3O+][HPO42-])/([H2PO4-])
74) Calculate the pH of a 0.020 M carbonic acid solution, H2CO3(aq), that has the stepwise dissociation
constants Ka1 = 4.3 × 10-7 and Ka2 = 5.6 × 10-11.
A) 1.70
B) 4.03
C) 6.37
D) 10.25
75) Calculate the pH of a 0.20 M H2SO3 solution that has the stepwise dissociation constants Ka1 = 1.5 ×
10-2 and Ka2 = 6.3 × 10-8.
A) 1.26
B) 1.32
C) 1.82
D) 2.52

76) Calculate the concentration of bicarbonate ion, HCO3-, in a 0.010 M H2CO3 solution that has the
stepwise dissociation constants Ka1 = 4.3 × 10-7 and Ka2 = 5.6 × 10-11.
A) 6.6 × 10-5 M
B) 4.3 × 10-7 M
C) 4.3 × 10-9 M
D) 5.6 × 10-11 M
77) What is the pH of a 0.10 M H2Se solution that has the stepwise dissociation constants Ka1 = 1.3 × 10-4
and Ka2 = 1.0 × 10-11?
A) 2.44
B) 3.89
C) 4.89
D) 5.50
78) What is the selenide ion concentration [Se2-] for a 0.100 M H2Se solution that has the stepwise
dissociation constants of Ka1 = 1.3 × 10-4 and Ka2 = 1.0 × 10-11?
A) 3.6 × 10-3 M
B) 1.3 × 10-4 M
C) 1.3 × 10-5 M
D) 1.0 × 10-11 M
79) Which of the following can be classified as a weak base?
A) CH3NH2
B) NH2OH
C) Both CH3NH2 and NH2OH
D) Neither CH3NH2 nor NH2OH

80) What is the pH of a 0.100 M NH3 solution that has Kb = 1.8 × 10-5? The equation for the dissociation of
NH3 is
NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq).
A) 1.87
B) 2.87
C) 11.13
D) 12.13
81) What is the pH of a 0.30 M pyridine solution that has a Kb = 1.9 × 10-9? The equation for the
dissociation of pyridine is
C5H5N(aq) + H2O(l) ⇌ C5H5NH+(aq) + OH-(aq).
A) 4.62
B) 8.72
C) 9.38
D) 10.38
82) Aniline, (C6H5NH2, Kb = 4.3 × 10-10 at 25°C) is an industrially important amine used in the making of
dyes. Determine the pH of an aniline solution made by dissolving 3.90 g of aniline in enough water to
make 100 mL of solution.
A) 4.87
B) 9.13
C) 9.74
D) 10.74

83) Determine the ammonia concentration of an aqueous solution that has a pH of 11.50. The equation for
the dissociation of NH3 (Kb = 1.8 × 10-5) is
NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq).
A) 2.5 M
B) 0.55 M
C) 5.7 × 10-3 M
D) 3.2 × 10-3 M
84) How many grams of pyridine are there in 100 mL of an aqueous solution that has a pH of 9.00? The
Kb for pyridine is 1.9 × 10-9 and the equation of interest is
C5H5N(aq) + H2O(l) ⇌ C5H5NH+(aq) + OH-(aq).
A) 0.053 g
B) 0.42 g
C) 0.79 g
D) 7.9 g
85) Ammonia NH3, has a base dissociation constant of 1.8 × 10-5. What is the conjugate acid of ammonia
and what is its acid dissociation constant?
A) NH4+, 1.9 × 109
B) NH4+, 1.8 × 10-5
C) NH4+, 5.6 × 10-10
D) NH2-, 5.6 × 10-10

86) Methylamine CH3NH2, has a base dissociation constant of 3.7 × 10-4. What is the conjugate acid of
methylamine and what is its acid dissociation constant?
A) CH3NH3+, 2.7 × 103
B) CH3NH3+, 3.7 × 10-4
C) CH3NH3+, 2.7 × 10-11
D) CH3NH2-, 2.7 × 10-11
87) Acetic acid CH3COOH, has an acid dissociation constant of 1.8 × 10-5. What is the conjugate base of
acetic acid and what is its base dissociation constant?
A) CH3C(OH)2+, 5.6 × 104
B) CH3C(OH)2+, 5.6 × 10-10
C) CH3COOH, 5.6 × 10-10
D) CH3CO2-, 5.6 × 10-10
88) Dihydrogen phosphate H2PO4-, has an acid dissociation constant of 6.2 × 10-7. What is the conjugate
base of H2PO4- and what is its base dissociation constant?
A) H3PO4, 1.6 × 106
B) H3PO4, 1.6 × 10-8
C) HPO42-, 1.6 × 106
D) HPO42-, 1.6 × 10-8

89) If you know Kb for ammonia, NH3, you can calculate the equilibrium constant, Ka, for the following
reaction:
NH4+ + H2O ⇌ NH3 + H3O+
using the equation:
A) Ka = Kw × Kb
B) Ka = Kw / Kb
C) Ka = 1 / Kb
D) Ka = Kb / Kw
90) If an equal number of moles of the weak acid HCN and the strong base KOH are added to water, is
the resulting solution acidic, basic, or neutral?
A) acidic
B) basic
C) neutral
D) There is insufficient information provided to answer this question.
91) Arrange the following 0.10 M aqueous solutions in order of increasing pH:
NaOH, HBr, NaCH3CO2, KBr, NH4Br.
A) HBr, KBr, NH4Br, NaCH3CO2, NaOH
B) NaOH, NaCH3CO2, NH4Br, KBr, HBr
C) NaOH, NaCH3CO2, KBr, NH4Br, HBr
D) HBr, NH4Br, KBr, NaCH3CO2, NaOH

92) Which one of the following salts, when dissolved in water, produces the solution with a pH closest to
7.00?
A) NH4Cl
B) BaO
C) NaHSO4
D) RbI
93) Calculate the pH of a 0.100 M NaCH3CO2 solution. Ka for acetic acid, CH3CO2H, is 1.8 × 10-5.
A) 2.87
B) 5.13
C) 8.87
D) 11.13
94) Calculate the pH of a of 0.100 M KBrO solution. Ka for hypobromous acid, HBrO, is 2.0 × 10-9.
A) 3.15
B) 4.85
C) 9.15
D) 10.85
95) Calculate the pH of a 0.100 M CH3NH3Cl solution. Kb for methylamine, CH3NH2, is 3.7 × 10-4.
A) 2.22
B) 5.78
C) 8.22
D) 11.78

96) Equal volumes of 0.10 M NH3 (Kb = 1.8 × 10-5) and 0.10 M HCN (Ka = 4.9 × 10-10) are mixed together.
Will the resulting solution be acidic, basic, or neutral?
A) acidic
B) basic
C) neutral
D) insufficient information to solve
97) What is the identity of M in the hydrate M(H2O)6n+ that has the 0.10 M solution with the lowest pH?
A) Li+
B) Na+
C) Mg2+
D) Al3+
98) What is the identity of M in the hydrate M(H2O)6n+ that has the 0.10 M solution with the highest pH?
A) Li+
B) Na+
C) Mg2+
D) Al3+
99) Identify the Lewis acid that acts as a reactant in the following reaction.
Fe(H2O)63+(aq) + 6 CN-(aq) → Fe(CN)63-(aq) + 6 H2O(l)
A) Fe3+
B) H2O
C) CN-
D) Fe(H2O)63+

100) Identify the Lewis acid that acts as a reactant in the following reaction.
Co(H2O)63+(aq) + 6 NH3(aq) → Co(NH3)63+(aq) + 6 H2O(l)
A) Co(H2O)63+
B) Co3+
C) NH3
D) H2O
101) For Cu2+ and CO2, which will behave as a Lewis acid toward OH- in water?
A) only Cu2+
B) only CO2
C) Cu2+ and CO2
D) neither Cu2+ nor CO2
102) Which one of the following is expected to be the strongest Lewis acid?
A) Fe
B) Fe+
C) Fe2+
D) Fe3+

103) Which of the following species cannot act as a Lewis base?
A) O2-
B) CH4
C) H2O
D) NH3
104) Identify the Br∅nsted-Lowry acids.
A) (1) and (3)
B) (1) and (4)
C) (2) and (3)
D) (2) and (4)
105) Identify the Br∅nsted-Lowry bases.
A) (1) and (3)
B) (1) and (4)
C) (2) and (3)
D) (2) and (4)
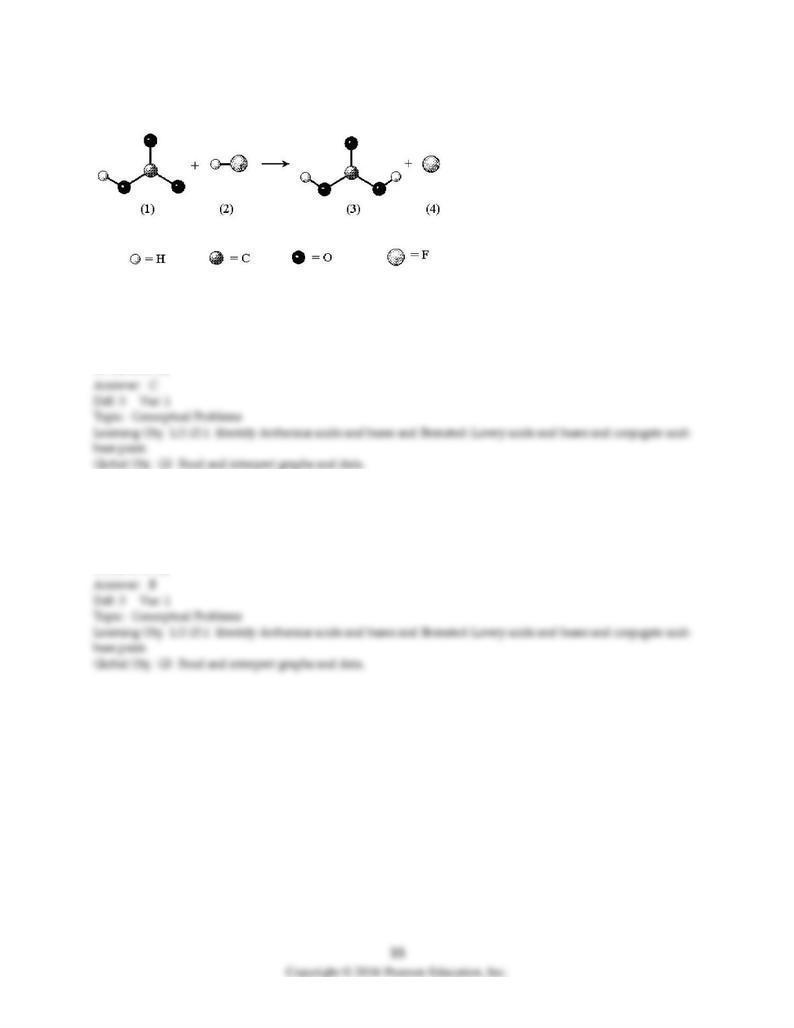
In the following reaction the unshaded spheres represent H atoms.
106) Identify the Br∅nsted-Lowry acids.
A) (1) and (3)
B) (1) and (4)
C) (2) and (3)
D) (2) and (4)
107) Identify the Br∅nsted-Lowry bases.
A) (1) and (3)
B) (1) and (4)
C) (2) and (3)
D) (2) and (4)
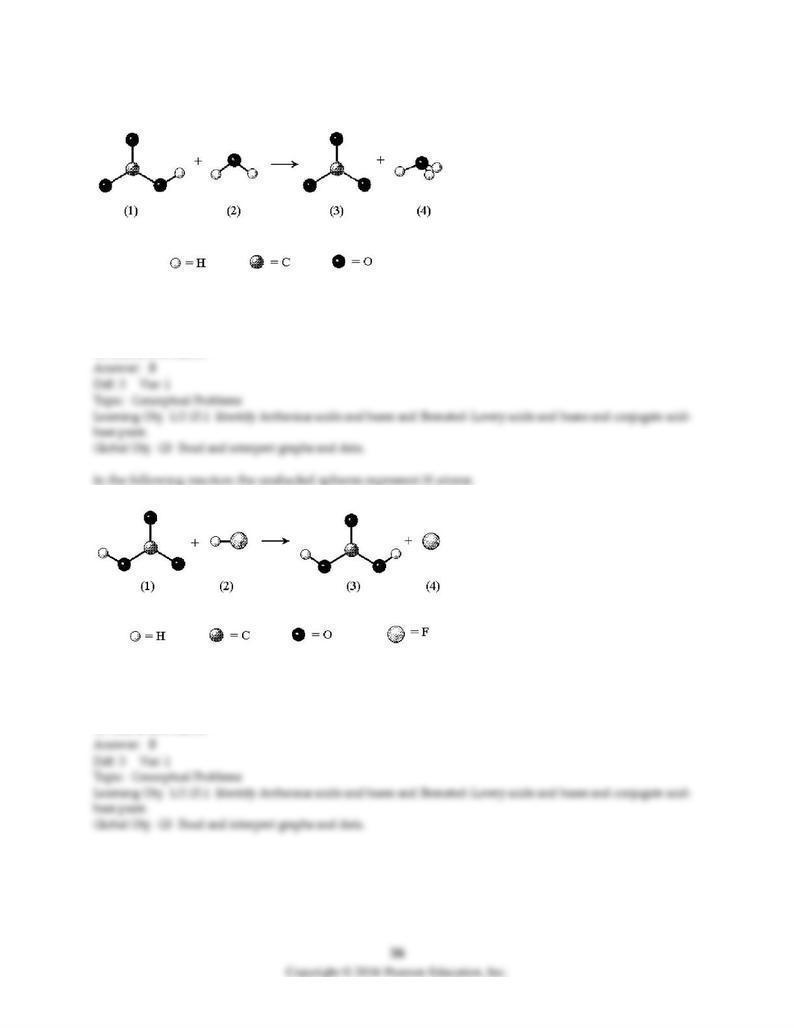
In the following reaction the unshaded spheres represent H atoms.
108) Identify the Br∅nsted-Lowry acid/base conjugate pairs.
A) (1)/(2) and (3)/(4)
B) (1)/(3) and (2)/(4)
C) (1)/(4) and (2)/(3)
109) Identify the Br∅nsted-Lowry acid/base conjugate pairs.
A) (1)/(2) and (3)/(4)
B) (1)/(3) and (2)/(4)
C) (1)/(4) and (2)/(3)

The following pictures represent aqueous solutions of three acids HA (A = X, Y, or Z); water molecules
110) Arrange the three acids in order of increasing acid strength.
A) HZ < HY < HX
B) HY < HZ < HX
C) HZ < HX < HY
D) HX < HZ < HY
111) Which acid, if any, is a strong acid?
A) All are strong acids.
B) HX and HZ
C) HY
D) None are strong acids.

112) Which acid has the smallest value of Ka?
A) HX
B) HY
C) HZ
D) All have the same Ka value.
113) Which acid has the lowest percent dissociation?
A) HX
B) HY
C) HZ
D) All have the same percent dissociation.
114) Which acid solution has the lowest pH?
A) HX
B) HY
C) HZ
D) All have the same pH.

115) Which of the above pictures represents a solution of a weak diprotic acid H2A for which Ka1 >> Ka2?
(Water molecules have been omitted for clarity.)
A) picture (a)
B) picture (b)
C) picture (c)
D) picture (d)
116) Which of the above pictures represents a solution of a diprotic acid H2A for which Ka1 = ∞ and Ka2
is exceptionally small. (Water molecules have been omitted for clarity.)
A) picture (a)
B) picture (b)
C) picture (c)
D) picture (d)

The following pictures represent equal volumes of aqueous solutions of three acids HA (A = X, Y, or Z);
117) Arrange the acids in order of increasing acid strength.
A) HZ < HY < HX
B) HY < HZ < HX
C) HZ < HX < HY
D) HX < HZ < HY
118) Arrange the acids in order of increasing value of Ka.
A) Ka(HZ) < Ka(HY) < Ka(HX)
B) Ka(HY) < Ka(HZ) < Ka(HX)
C) Ka(HZ) < Ka(HX) < Ka(HY)
D) Ka(HX) < Ka(HZ) < Ka(HY)
119) Arrange the acids in order of increasing value of percent dissociation.
A) HZ < HY < HX
B) HY < HZ < HX
C) HZ < HX < HY
D) HX < HZ < HY

120) Arrange the acids in order of increasing pH.
A) HZ < HY < HX
B) HY < HZ < HX
C) HZ < HX < HY
D) HX < HZ < HY
121) Arrange the conjugate bases (A- = X-, Y-, or Z-) in order of increasing base strength.
A) Z- < Y- < X-
B) Y- < Z- < X-
C) Z- < X- < Y-
D) X- < Z- < Y-
122) Arrange the conjugate bases (A- = X-, Y-, or Z-) in order of increasing value of Kb.
A) Kb(Z-) < Kb(Y-) < Kb(X-)
B) Kb(Y-) < Kb(Z-) < Kb(X-)
C) Kb(Z-) < Kb(X-) < Kb(Y-)
D) Kb(X-) < Kb(Z-) < Kb(Y-)
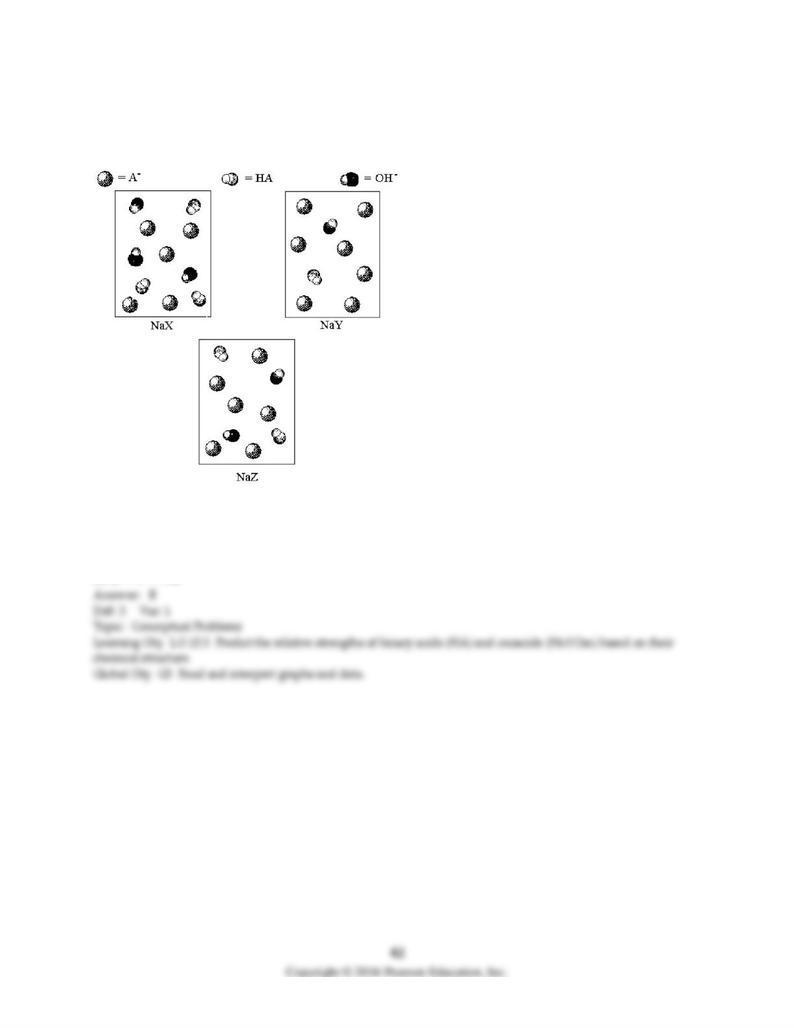
123) The following pictures represent solutions of three salts NaA (A- = X-, Y-, or Z-); water molecules
and Na+ ions have been omitted for clarity. Arrange the three A- ions in order of decreasing base
strength.
A) X- > Y- > Z-
B) X- > Z- > Y-
C) Y- > Z- > X-
D) Z- > Y- > XZ-

The following pictures represent solutions of three salts MA; water molecules have been omitted for
124) Which picture represents an acidic salt?
A) (1)
B) (2)
C) (3)
D) None of these
125) Which picture represents a neutral salt?
A) (1)
B) (2)
C) (3)
D) None of these
126) Which picture represents a basic salt?
A) (1)
B) (2)
C) (3)
D) None of these

127) Of the elements indicated on the periodic table shown above, which forms the strongest binary acid,
H2X or HX, where
A) A
B) B
C) C
D) D
128) Of the elements indicated on the periodic table shown above, which forms the weakest binary acid,
H2X or HX, where X = A, B, C, or D?
A) A
B) B
C) C
D) D
129) Of the elements indicated on the periodic table shown above, which forms the strongest oxoacid acid
with the formula H2XO3 or HXO3, where X = A, B, C, or D?
A) A
B) B
C) C
D) D
130) Of the elements indicated on the periodic table shown above, which forms the weakest oxoacid acid
with the formula H2XO3 or HXO3, where X = A, B, C, or D?
A) A
B) B
C) C
D) D
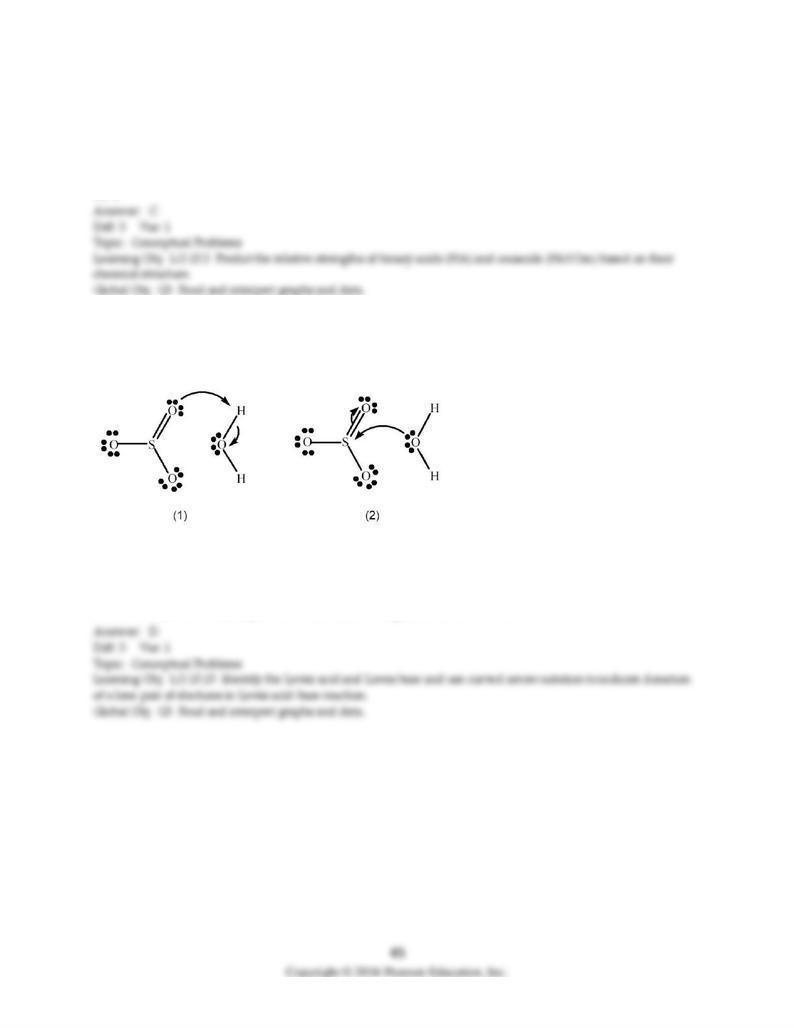
131) SO3 reacts with H2O to form H2SO4. Which picture below correctly represents the curved arrow
notation for the initial Lewis acid-Lewis base interaction in this reaction; what is the Lewis acid and the
Lewis base?
A) Picture (1) is correct; H2O is the Lewis acid and SO3 is the Lewis base.
B) Picture (1) is correct; SO3 is the Lewis acid and H2O is the Lewis base.
C) Picture (2) is correct; H2O is the Lewis acid and SO3 is the Lewis base.
D) Picture (2) is correct; SO3 is the Lewis acid and H2O is the Lewis base.

132) CO2 reacts with H2O to form HCO3- and H+. Which picture below correctly represents the curved
arrow notation for the initial Lewis acid-Lewis base interaction in this reaction; what is the Lewis acid
and the Lewis base?
(1) (2)
A) Picture (1) is correct; H2O is the Lewis acid and CO2 is the Lewis base.
B) Picture (1) is correct; CO2 is the Lewis acid and H2O is the Lewis base.
C) Picture (2) is correct; H2O is the Lewis acid and CO2 is the Lewis base.
D) Picture (2) is correct; CO2 is the Lewis acid and H2O is the Lewis base.
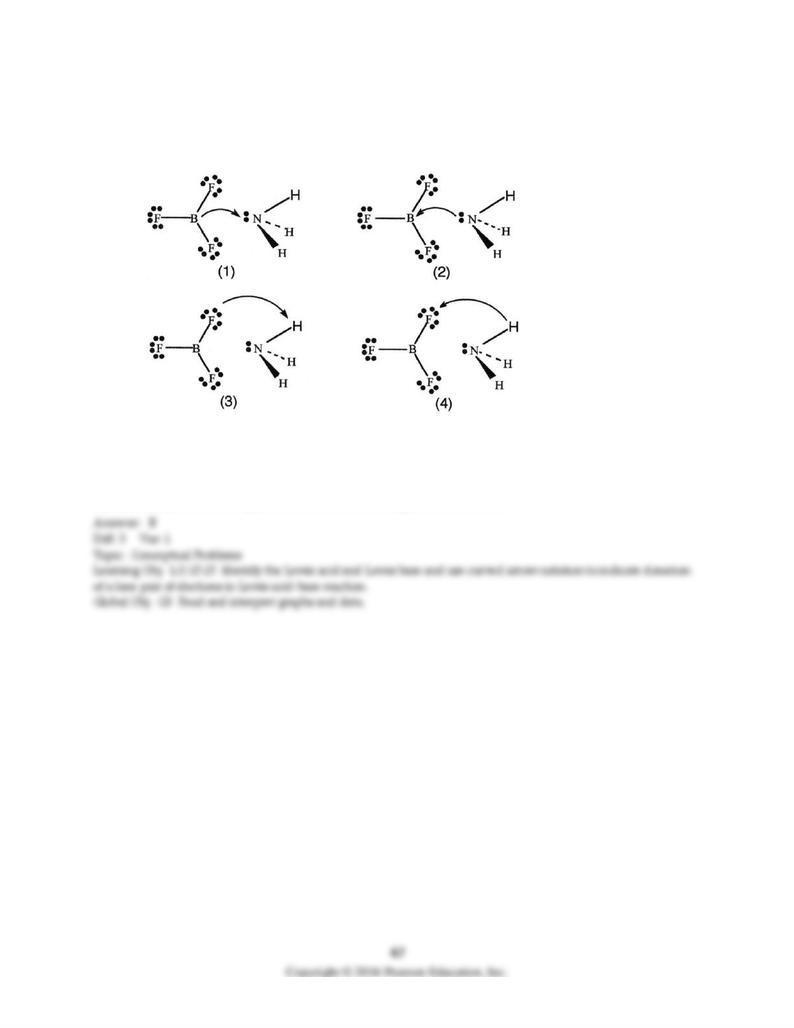
133) BF3 and NH3 undergo a Lewis acid-base reaction forming an adduct. Which picture below correctly
represents the curved arrow notation for the initial Lewis acid-Lewis base interaction in this reaction;
what is the Lewis acid and the Lewis base?
A) Picture (1) is correct; NH3 is the Lewis acid and BF3 is the Lewis base.
B) Picture (2) is correct; BF3 is the Lewis acid and NH3 is the Lewis base.
C) Picture (3) is correct; NH3 is the Lewis acid and BF3 is the Lewis base.
D) Picture (4) is correct; BF3 is the Lewis acid and NH3 is the Lewis base.

15.2 Algorithmic Questions
1) When dissolved in water, which compound is generally considered to be an Arrhenius acid?
A) CH3CO2H
B) NaOH
C) Na2CO3
D) CH3CH2OH
2) Which Br∅nsted-Lowry acid is not considered to be a strong acid in water?
A) HCl
B) HI
C) H ClO3
D) H2SO4
3) An Arrhenius acid is best defined as a
A) electron pair donor.
B) proton acceptor.
C) substance that dissociates in water to produce aqueous hydrogen ions.
D) substance that dissociates in water to produce aqueous hydroxide ions.
4) An Arrhenius base is best defined as a
A) electron pair donor.
B) hydroxide acceptor.
C) substance that dissociates in water to produce aqueous hydrogen ions.
D) substance that dissociates in water to produce aqueous hydroxide ions.

5) When dissolved in water, which of the following compounds is an Arrhenius base?
A) CH3OH
B) HOCl
C) NaOH
D) CsF
6) A Br∅nsted-Lowry acid is best defined as a substance that can
A) accept an hydroxide ion.
B) donate an hydroxide ion.
C) accept a proton.
D) donate a proton.
7) Which one of the following species acts as a Br∅nsted-Lowry acid in water?
A) NaH
B) NH4+
C) CH3NHCH3
D) C6H6

8) In the following chemical equation indicate the reactant that is a Br∅nsted-Lowry base.
HCN(aq) + H2O(l) ⇌ H3O+(aq) + CN-(aq)
A) HCN
B) H2O
C) H3O+
D) CN-
9) Indicate all the Br∅nsted-Lowry acids in the following chemical reaction.
HBr(aq) + H2O(aq) ⇌ H3O+(aq) + Br-(aq)
A) HBr, H2O
B) HBr, H3O+
C) HBr, Br-
D) HBr, H2O, H3O+
10) Which one of the following can behave either as a Br∅nsted-Lowry acid or a Br∅nsted-Lowry base in
an aqueous solution reaction?
A) HSO4-
B) CH4
C) HI
D) H3PO4

11) Which Br∅nsted-Lowry base has the strongest conjugate acid?
A) CH3CO2-
B) OH-
C) F-
D) NO3-
12) Which Br∅nsted-Lowry acid has the strongest conjugate base?
A) CH4
B) HClO4
C) HF
D) Hl
13) What is the geometric shape of the hydrated proton; that is, the hydronium ion H3O+?
A) angular
B) trigonal pyramidal
C) trigonal planar
D) tetrahedral
14) Which is not a hydrate of a proton?
A) H3O+
B) H17O8+
C) H19O8+
D) H43O21+

15) What is the strongest acid among the following?
A) HF
B) HCl
C) H2O
D) H2S
16) Which acid of the following set has the strongest conjugate base?
A) CH4
B) NH3
C) H2S
D) HCl
17) What is the strongest base among the following?
A) IO-
B) IO2-
C) IO3-
D) IO4-
18) What is the strongest acid among the following?
A) H2SO3
B) H2SO4
C) H2SeO2
D) H2TeO4

19) What is the strongest acid among the following?
A) HIO
B) HIO2
C) HIO3
D) HIO4
20) What is the strongest acid among the following?
A) CH3CO2H
B) ClCH2CO2H
C) Cl2CHCO2H
D) Cl3CCO2H
21) Calculate the hydronium ion concentration in an aqueous solution that contains 2.50 × 10-4 M in
hydroxide ion.
A) 4.00 × 10-9 M
B) 4.00 × 10-10 M
C) 4.00 × 10-11 M
D) 5.00 × 10-11 M
22) Calculate the hydroxide ion concentration in an aqueous solution that contains 3.50 × 10-5 M in
hydronium ion.
A) 2.86 × 10-2 M
B) 2.86 × 10-9 M
C) 2.86 × 10-10 M
D) 3.50 × 10-10 M

23) A solution with a hydrogen ion concentration of 3.25 × 10-5 M is ________ and has a hydroxide ion
concentration of ________.
A) acidic, 3.08 × 10-9 M
B) acidic, 3.08 × 10-10 M
C) basic, 3.08 × 10-9 M
D) basic, 3.08 × 10-10 M
24) A solution with a hydroxide ion concentration of 4.15 × 10-7 M is ________ and has a hydrogen ion
concentration of ________.
A) acidic, 2.41 × 10-7 M
B) acidic, 2.41 × 10-8 M
C) basic, 2.41 × 10-7 M
D) basic, 2.41 × 10-8 M
25) Normal rainfall has a concentration of OH- that is 3.99 × 10-9. The concentration of H3O+ in normal
rainfall is
A) greater than 3.99 × 10-9, and the rain is acidic.
B) greater than 3.99 × 10-9, and the rain is basic.
C) less than 3.99 × 10-9, and the rain is acidic.
D) less than 3.99 × 10-9, and the rain is basic.
26) Calculate the pH for an aqueous solution of acetic acid that contains hydronium ion.
A) 2.41 × 10-12 M
B) 4.15 × 10-3 M
C) 2.38
D) 11.62

27) Calculate the pH for an aqueous solution of pyridine that contains hydroxide ion.
A) 2.41 × 10-11
B) 4.15 × 10-4
C) 3.38
D) 10.62
28) What is the hydroxide ion concentration and the pH for a hydrochloric acid solution that has a
hydronium ion concentration of
A) 6.67 × 10-8 M, 6.82
B) 6.67 × 10-8 M, 7.18
C) 6.67 × 10-9 M, 5.82
D) 6.67 × 10-9 M, 8.17
29) What is the hydronium ion concentration and the pH for an aqueous solution of NH3 that has a
hydroxide ion concentration of
A) 4.44 × 10-10 M and 4.65
B) 4.44 × 10-10 M and 9.35
C) 4.44 × 10-11 M and 3.65
D) 4.44 × 10-11 M and 10.35
30) What is the hydronium ion concentration of an acid rain sample that has a pH of 3.25?
A) 1.78 × 10-11 M
B) 5.62 × 10-4 M
C) 3.25 M
D) 10.75 M

31) What is the hydroxide ion concentration of a lye solution that has a pH of 13.20?
A) 6.31 × 10-14 M
B) 1.58 × 10-1 M
C) 0.80 M
D) 13.20 M
32) What statement is most consistent for an acid with a pH = 1?
A) one one-hundredth as strong as an acid with a pH of 3
B) half a strong as an acid with a pH = 3
C) twice as strong as an acid with a pH of 3
D) one hundred times as strong as an acid with a pH = 3
33) What is the pH of a 0.040 M HClO4 solution?
A) 0.040
B) 0.080
C) 1.40
D) 12.60
34) What is the pH of a 0.020 M Ba(OH)2 solution?
A) 1.40
B) 1.70
C) 12.30
D) 12.60

35) What is the pH of a solution prepared by dissolving 0.30 grams of solid CaO (lime) in enough water to
make 2.00 L of aqueous Ca(OH)2 (limewater)?
CaO(s) + H2O(l) → Ca2+(aq) + 2 OH-(aq)
A) 2.27
B) 2.57
C) 11.42
D) 11.73
36) What is the pH of a solution prepared by diluting 75.00 mL of 0.10 M HCl with enough water to
produce a total volume of 100.00 mL?
A) 1.00
B) 1.12
C) 2.00
D) 2.24
37) What is the pH of a solution prepared by diluting 50.00 mL of 0.020 M Ba(OH)2 with enough water to
produce a total volume of 250.00 mL?
A) 2.10
B) 2.40
C) 11.60
D) 11.90

38) What is the pH of a solution prepared by mixing 100.00 mL of 0.20 M HCl with 50.00 mL of 0.30 M
HCl? Assume that the volumes are additive.
A) 0.25
B) 0.30
C) 0.63
D) 1.70
39) What is the pH of a solution prepared by mixing 100.00 mL of 0.020 M Ca(OH)2 with 50.00 mL of
0.200 M NaOH? Assume that the volumes are additive.
A) 12.90
B) 12.97
C) 13.15
D) 13.45
40) What is the pH of a 0.020 M RbOH solution?
A) 0.020
B) 0.040
C) 1.70
D) 12.30
41) What is the pH of a solution prepared by mixing 100.00 mL of 0.025 M Ca(OH)2 with 50.00 mL of
0.080 M LiOH? Assume that the volumes are additive.
A) 12.67
B) 12.78
C) 12.95
D) 13.25

42) What is the hydronium ion concentration of a 0.300 M acetic acid solution with Ka = 1.8 × 10-5? The
equation for the dissociation of acetic acid is:
CH3CO2H(aq) + H2O(l) ⇌ H3O+(aq) + CH3CO2-(aq).
A) 2.3 × 10-2 M
B) 4.2 × 10-2 M
C) 2.3 × 10-3 M
D) 4.2 × 10-3 M
43) What is the hydronium ion concentration of a 0.200 M hypochlorous acid solution with
The equation for the dissociation of hypochlorous acid is:
HOCl(aq) + H2O(l) ⇌ H3O+(aq) + OCl-(aq).
A) 1.9 × 10-4 M
B) 8.4 × 10-4 M
C) 3.7 × 10-5 M
D) 8.4 × 10-5 M
44) Potassium hydrogen phthalate (molar mass = 204.2 g/mol)is one of the most commonly used acids for
standardizing solutions containing bases. KHP is a monoprotic weak acid with Calculate
the pH of the solution that results when 0.40 g of KHP is dissolved in enough water to produce 25.0 mL
of solution.
A) 2.10
B) 3.26
C) 4.30
D) 5.41

45) A 0.50 M solution of a weak acid HA has the same pH as a 0.075 M solution of HCl. Calculate the Ka
for HA.
A) 0.50
B) 0.075
C) 0.1324
D) 0.01125
46) A weak base ionizes in water as follows:
B(aq) + H2O(l) ⇌ BH+(aq) + OH-(aq)
A 0.150 M solution of B is 2.500 % ionized at 25ºC. What is the pH of this solution?
A) 0.40
B) 2.43
C) 11.57
D) 13.60
47) Calculate the pH of a 0.060 M carbonic acid solution, H2CO3(aq), that has the stepwise dissociation
constants Ka1 = 4.3 × 10-7 and Ka2 = 5.6 × 10-11.
A) 1.22
B) 3.79
C) 6.37
D) 10.25

48) Calculate the pH of a 0.20 M H2SO3, solution that has the stepwise dissociation constants Ka1 = 1.5 ×
10-2 and Ka2 = 6.3 × 10-8.
A) 1.26
B) 1.32
C) 1.82
D) 2.52
49) Calculate the concentration of bicarbonate ion, HCO3-, in a 0.050 M H2CO3 solution that has the
stepwise dissociation constants Ka1 = 4.3 × 10-7 and Ka2 = 5.6 × 10-11.
A) 1.5 × 10-4 M
B) 4.3 × 10-7 M
C) 2.2 × 10-8 M
D) 5.6 × 10-11 M
50) What is the pH of a 0.20 M H2Se solution that has the stepwise dissociation constants Ka1 = 1.3 × 10-4
and Ka2 = 1.0 × 10-11?
A) 2.29
B) 3.89
C) 4.59
D) 5.57
51) What is the selenide ion concentration [Se2-] for a 0.400 M H2Se solution that has the stepwise
dissociation constants of Ka1 = 1.3 × 10-4 and Ka2 = 1.0 × 10-11?
A) 7.2 × 10-3 M
B) 1.3 × 10-4 M
C) 5.2 × 10-5 M
D) 1.0 × 10-11 M

52) Which of the following is a weak diprotic acids?
A) HCN
B) H CO2H
C) H2CO3
D) H2SO3
53) What is the pH of a 0.300 M NH3 solution that has Kb = 1.8 × 10-5? The equation for the dissociation of
NH3 is
NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq).
A) 2.11
B) 2.63
C) 11.89
D) 11.37
54) What is the pH of a 2.4 M pyridine solution that has Kb = 1.9 × 10-9? The equation for the dissociation
of pyridine is
C5H5N(aq) + H2O(l) ⇌ C5H5NH+(aq) + OH-(aq).
A) 4.17
B) 8.72
C) 9.83
D) 10.83

55) Aniline, (C6H5NH2, Kb = 4.3 × 10-10 at 25°C) is an industrially important amine used in the making of
dyes. Determine the pH of an aniline solution made by dissolving 3.90 g of aniline in enough water to
make 100 mL of solution.
A) 4.87
B) 9.13
C) 9.74
D) 10.74
56) Determine the ammonia concentration of an aqueous solution that has a pH of 11.30. The equation for
the dissociation of NH3 (Kb = 1.8 × 10-5) is
NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq).
A) 2.7 M
B) 0.22 M
C) 9.0 × 10-3 M
D) 2.0 × 10-3 M
57) What is the relationship between Ka and Kb at 25°C for a conjugate acid base pair?
A) Ka × Kb = 1 × 10-14
B) Ka/Kb = 1 × 10-14
C) Kb/Ka = 1 × 10-14
D) Ka - Kb = 1 × 10-14

58) Which one of the following salts, when dissolved in water, produces the solution with the highest
pH?
A) KHSO4
B) RbClO4
C) BaO
D) CH3CH3NH3Br
59) If an equal number of moles of the weak acid HF and the strong base KOH are added to water, is the
resulting solution acidic, basic, or neutral?
A) acidic
B) basic
C) neutral
D) There is insufficient information provided to answer this question.
60) Which one of the following salts, when dissolved in water, produces the solution with the highest
pH?
A) NaI
B) KBr
C) RbCl
D) CsF
61) Which one of the following salts, when dissolved in water, produces the solution with a pH closest to
7.00?
A) NH4I
B) Na2O
C) KHCO3
D) CsCl

62) Calculate the pH of a 0.200 M NaCH3CO2 solution. Ka for acetic acid, CH3CO2H, is 1.8 × 10-5.
A) 2.72
B) 4.98
C) 9.02
D) 11.28
63) Calculate the pH of a 0.800 M KBrO solution. Ka for hypobromous acid, HBrO, is 2.0 × 10-9.
A) 2.70
B) 4.40
C) 9.60
D) 11.30
64) Calculate the pH of a 0.100 M CH3NH3Cl solution. Kb for methylamine, CH3NH2, is 3.7 × 10-4.
A) 2.22
B) 5.78
C) 8.22
D) 11.78
65) Which one of the following salts, when dissolved in water, produces the solution with the lowest pH?
A) NaI
B) KI
C) MgI2
D) AlI3

66) Which of the following salts are acidic?
A) LiCl, NaCl, KCl
B) NH4Cl, CuCl2, AlCl3
C) NaCH3CO2, LiCH3CO2, RbCH3CO2
D) KCl, NH4Cl, Na2CO3
67) Which one of the following salts, when dissolved in water, produces the solution with the highest
pH?
A) LiHSO4
B) NaClO4
C) KF
D) CH3NH3I
68) Which one of the following salts, when dissolved in water, produces the solution with the highest
pH?
A) LiI
B) LiBr
C) LiCl
D) LiF
69) Which one of the following salts, when dissolved in water, produces the solution with the lowest pH?
A) RbCl
B) NH4Cl
C) SrCl2
D) AlCl3

70) Which one of the following is not considered to be a Lewis base?
A) H2S
B) NH3
C) NH4+
D) Cl-
71) Which one of the following is least able to behave as a Lewis base?
A) CH3NH2
B) (CH3)2NH
C) (CH3)3N
D) (CH3)2NCH2CH3+
72) The compound BCl3 can be described as a(n)
A) Arrhenius acid.
B) Br∅nsted-Lowry base.
C) Lewis acid.
D) Lewis base.
73) Identify the set of Lewis acids.
A) BH3, BF3, Cu2+, CO2
B) Cl-, OH-, NBr3, H2O
C) H3PO4, H2PO4-, HPO42-, PO43-
D) CCl3-, NH2-, CN-, F-

15.3 Short Answer Questions
1) The Br∅nsted-Lowry acids in the chemical equation below are ________ and ________.
HNO2(aq) + H2O(l) ⇌ H3O+(aq) + NO2-(aq)
2) The equilibrium constant for the reaction below has the value Ka = 3.5 × 10-4. In this reaction the
stronger Br∅nsted-Lowry acid is ________ and the weaker Br∅nsted-Lowry acid is ________.
HF(aq) + H2O(l) ⇌ H3O+(aq) + F-(aq)
3) A proton hydrated by ten water molecules has the formula ________.
4) The number of waters of hydration in the hydrate proton H11O5+ is ________.
5) A solution with hydronium ion concentration [H+] = 1.60 × 10-2 M has a hydroxide ion concentration
[OH-] = ________.

6) The concentration of H3O+ in human sweat can be as low as 2.5 × 10-6. The concentration of OH- in the
sweat is ________, and this solution is ________ (acidic, basic, neutral).
7) Undersea flora prefer a maximum concentration of OH- of 1.58 × 10-5. The concentration of H3O+ in
the seawater is ________, and this solution is ________ (acidic, basic, neutral).
8) At the normal body temperature of 37°C, Kw = 2.42 × 10-14. The H3O+ concentration of normal blood
ranges from 3.5 × 10-8 to 4.5 × 10-8. The OH- concentration of normal blood ranges from ________ to
________, and blood is ________ (acidic, basic, neutral).
9) At 50°C the value of Kw is 5.5 × 10-14, and the pH of a neutral solution at 50°C is ________.
10) One way to prepare a solution with a pH of 10.00 is to dissolve ________ grams of CaO in enough
water to make 500 mL of solution.
11) Bromothymol blue indicator changes color from yellow at a pH of 6.0 to blue at a pH of 7.6. Phenol
red indicator changes color from yellow at a pH of 6.8 to red at a pH of 8.4. A sample of pancreatic fluid
having [OH-] = 1.258 × 10-6 would impart a ________ color to bromothymol blue and a ________ color to
phenol red.

12) Bromothymol blue indicator changes color from yellow at a pH of 6.0 to blue at a pH of 7.6. Methyl
red indicator changes color from red at a pH of 4.4 to yellow at a pH of 6.2. A sample of saliva having
[H3O+] = 6.310 × 10-7 would impart a ________ color to bromothymol blue and a ________ color to methyl
red.
13) The pH of a 0.055 M KOH solution is ________.
14) The value of Ka for a 0.250 M HCN solution having a pH of 4.956 is ________.
15) In order for the reaction HA + HSO3- ⇌ A- + H2SO3 to have an equilibrium constant Kc < 1, the Ka of
HA must be ________ (greater, less) than the Ka of HSO3-.
16) In order for the reaction A- + H2CO3 ⇌ HA + HCO3- to have an equilibrium constant Kc < 1, the Ka of
HA must be ________ (greater, less) than the Ka of H2CO3.

17) A 1.25 × 10-4 M solution of the anti-inflammatory drug naproxen has a pH = 4.2. The Ka and pKa of
naproxen are ________ and ________.
18) Phenobarbital is an antiepileptic drug with a water solubility of 4.3 × 10-3 M and pKa = 7.4. What is
the pH and percent ionization of 4.3 × 10-3 M phenobarbital?
19) Phenobarbital has a pKa = 7.4. Compared to a 1.0 × 10-3 M solution, 1.0 × 10-4 M phenobarbital will
have a ________ (higher, lower) pH and a ________ (higher, lower) percent ionization.
20) Erythromycin is a basic antimicrobial with pKb = 5.2. A 1.0 × 10-3 M solution of erythromycin has a
pH of ________ and a ________ percent ionization.
21) A 0.050 M solution of hydroxylamine, NH2OH, having Kb = 9.1 × 10-9 has a pH of ________.
22) A 0.50 M KNO2 solution will have a pH ________ seven.

23) The acid strength of an oxoacid having the general formula HnYOm increases as the electronegativity
of Y ________ and as the oxidation number of Y ________.
24) In the aquation reaction Co2+ + 6 H2O → [Co(H2O)6]2+, the Lewis acid is ________ and the Lewis
base is ________.
25) In the reaction between NO2 and H2O that produces HNO3, the Lewis acid is ________ and the Lewis
base is ________.