
Chemistry, 7e (McMurry/Fay)
Chapter 2 Atoms, Molecules, and Ions
2.1 Multiple-Choice Questions
1) What is the chemical symbol for manganese?
A) Hg
B) Mg
C) Mn
D) Na
2) Which element has the chemical symbol, P?
A) lead
B) phosphorus
C) platinum
D) potassium
3) According to history, the concept that all matter is composed of atoms was first proposed by
A) the Greek philosopher Democritus, but not widely accepted until modern times.
B) Dalton, but not widely accepted until the work of Mendeleev.
C) Dalton, but not widely accepted until the work of Einstein.
D) Dalton, and widely accepted within a few decades.
4) Mendeleev arranged the elements according to
A) atomic number and atomic weight.
B) atomic weight and chemical reactivity.
C) electron configuration and atomic weight.
D) physical state and relative abundance.

5) Which is not true?
A) Mendeleev ended each row in his periodic table with an inert gas.
B) Mendeleev left gaps in his periodic table for undiscovered elements.
C) Mendeleev ordered the elements in his periodic table by atomic weight.
D) Mendeleev's periodic table predated the concept of electron configuration.
6) The horizontal rows of the periodic table are called
A) groups.
B) periods.
C) triads.
D) elements.
7) The vertical columns of the periodic table are called
A) groups.
B) periods.
C) triads.
D) elements.
8) Most elements in the periodic table are
A) metals.
B) non-metals.
C) noble gases.
D) semi-metals.

9) Elements in a periodic group have similar
A) chemical properties.
B) densities.
C) masses.
D) physical properties.
10) Which horizontal row of the periodic table contains the most elements?
A) row 4
B) row 5
C) row 6
D) They all contain the same number of elements.
11) Which of the following statements does not describe a physical property of chlorine?
A) Chlorine combines with sodium to form table salt.
B) The color of chorine gas is green.
C) The density of chlorine gas at standard temperature and pressure is 3.17 g/L.
D) The freezing point of chlorine is -101°C.
12) Which of the following statements does not describe a chemical property of oxygen?
A) Iron will rust in the presence of oxygen.
B) Oxygen combines with carbon to form carbon dioxide gas.
C) The pressure is caused by collision of oxygen molecules with the sides of a container.
D) When coal is burned in oxygen, the process is called combustion.

13) Which group 5A element is most metallic?
A) N
B) P
C) Sb
D) Bi
14) Which group of elements reacts violently with water?
A) halogens
B) noble gases
C) alkali metals
D) alkaline earth metals
15) Gaseous elements characterized by low reactivity are found in group ________ of the periodic table.
A) 5A
B) 6A
C) 7A
D) 8A
16) The observation that 15.0 g of hydrogen reacts with 120.0 g of oxygen to form 135.0 g of water is
evidence for the law of
A) definite proportions.
B) energy conservation.
C) mass conservation.
D) multiple proportions.

17) The observation that 4.0 g of hydrogen reacts with 32.0 g of oxygen to form a product with O:H mass
ratio = 8:1, and 6.0 g of hydrogen reacts with 48.0 g of oxygen to form the same product with O/H mass
ratio = 8:1 is evidence for the law of
A) definite proportions.
B) energy conservation.
C) mass conservation.
D) multiple proportions.
18) Methane and oxygen react to form carbon dioxide and water. What mass of water is formed if 3.2 g of
methane reacts with 12.8 g of oxygen to produce 8.8 g of carbon dioxide?
A) 7.2 g
B) 8.8 g
C) 14.8 g
D) 16.0 g
19) Sodium metal and water react to form hydrogen and sodium hydroxide. If 5.98 g of sodium react with
water to form 0.26 g of hydrogen and 10.40 g of sodium hydroxide, what mass of water was consumed in
the reaction?
A) 4.68 g
B) 5.98 g
C) 10.14 g
D) 10.66 g

20) A sample of pure lithium carbonate contains 18.8% lithium by mass. What is the % lithium by mass in
a sample of pure lithium carbonate that has twice the mass of the first sample?
A) 9.40%
B) 18.8%
C) 37.6%
D) 75.2%
21) A sample of pure calcium fluoride with a mass of 15.0 g contains 7.70 g of calcium. How much
calcium is contained in 45.0 g of calcium fluoride?
A) 2.56 g
B) 7.70 g
C) 15.0 g
D) 23.1 g
22) The observation that hydrogen and oxygen can react to form two compounds with different chemical
and physical properties, one having an O:H mass ratio = 8:1 and the other having an O:H mass ratio =
16:1 is consistent with the law of
A) definite proportions.
B) energy conservation.
C) mass conservation.
D) multiple proportions.
23) Which of the following statements is not a postulate of Dalton's atomic theory?
A) Each element is characterized by the mass of its atoms.
B) Atoms are composed of protons, neutrons, and electrons.
C) Chemical reactions only rearrange atomic combinations.
D) Elements are composed of atoms.

24) Which of the following is a part of Dalton's atomic theory?
A) Atoms are rearranged but not changed during a chemical reaction.
B) Atoms break down during radioactive decay.
C) Atoms contain protons, neutrons, and electrons.
D) Isotopes of the same element have different masses.
25) Which of the following is not explained by Dalton's atomic theory?
A) conservation of mass during a chemical reaction
B) the existence of more than one isotope of an element
C) the law of definite proportions
D) the law of multiple proportions
26) Elements A and Q form two compounds, AQ and A2Q3. The mass ratio (mass Q)/(mass A) for AQ is
0.574. What is the mass ratio (mass Q)/(mass A) for A2Q3?
A) 0.383
B) 0.861
C) 1.16
D) 2.61
27) Elements A and Q form two compounds, AQ and A2Q. Which of the following must be true?
A) (mass Q)/(mass A) is one for AQ, and 1/2 for A2Q.
B) (mass Q)/(mass A) for AQ must equal (mass Q)/(mass A) for A2Q.
C) (mass Q)/(mass A) for AQ must be 2 times (mass Q)/(mass A) for A2Q.
D) (mass Q)/(mass A) for AQ must be 1/2 (mass Q)/(mass A) for A2Q.

28) Elements A and Q form two compounds. The ratio (mass Q)/(mass A) for compound one is 0.271 and
ratio (mass Q)/(mass A) for compound two is 0.362. If compound one has the chemical formula AQ, what
is the chemical formula for compound two?
A) A3Q4
B) A2Q3
C) AQ2
D) AQ3
29) The existence of electrons in atoms of all elements was demonstrated by
A) Millikan's oil drop experiment.
B) Rutherford's gold foil experiment.
C) Thomson's cathode ray tube experiment.
D) None of these
30) The charge-to-mass ratio of an electron was established by
A) Millikan's oil drop experiment.
B) Rutherford's gold foil experiment.
C) Thomson's cathode ray tube experiment.
D) None of these
31) The current model of the atom in which essentially all of an atom's mass is contained in a very small
nucleus, whereas most of an atom's volume is due to the space in which the atom's electrons move was
established by
A) Millikan's oil drop experiment.
B) Rutherford's gold foil experiment.
C) Thomson's cathode ray tube experiment.
D) None of these

32) The existence of neutrons in the nucleus of an atom was demonstrated by
A) Millikan's oil drop experiment.
B) Rutherford's gold foil experiment.
C) Thomson's cathode ray tube experiment.
D) None of these
33) Most of the alpha particles directed at a thin gold foil in Rutherford's experiment
A) bounced directly back from the foil.
B) passed directly through the foil undeflected.
C) passed through the foil but were deflected at an angle.
D) were absorbed by the foil.
34) Which subatomic particle has the smallest mass?
A) a proton
B) a neutron
C) an electron
D) an alpha particle
35) The symbol that is usually used to represent atomic number is
A) A.
B) N.
C) X.
D) Z.

36) The mass number of an atom is equal to the number of
A) electrons.
B) neutrons.
C) protons.
D) protons plus neutrons.
37) Which of the following two atoms are isotopes?
A) Ar and Ca
B) C and C
C) Cl and Br
D) Mg and C
38) Which of the following represent isotopes?
A: [ ] B: [ ] C: [ ] D: [ ]
A) A and B
B) A and C
C) A and D
D) C and D

39) Boron-9 can be represented as
A) Be.
B) B.
C) B.
D) B.
40) How many protons (p) and neutrons (n) are in an atom of Sr?
A) 38 p, 52 n
B) 38 p, 90 n
C) 52 p, 38 n
D) 90 p, 38 n
41) How many protons (p) and neutrons (n) are in an atom of calcium-46?
A) 20 p, 26 n
B) 20 p, 46 n
C) 26 p, 20 n
D) 46 p, 60 n

42) What is the chemical symbol for an atom that has 29 protons and 36 neutrons?
A) Cu
B) Kr
C) N
D) Tb
43) How many electrons are in a neutral atom of iodine-131?
A) 1
B) 53
C) 54
D) 131
44) How many protons (p), neutrons (n), and electrons (e) are in one atom of Mg?
A) 12 p, 12 n, 12 e
B) 12 p, 11 n, 12 e
C) 12 p, 11 n, 10 e
D) 12 p, 11 n, 14 e
45) Identify the chemical symbol of element Q in Q.
A) Br
B) Hg
C) Pd
D) Se

46) The atoms of a particular element all have the same number of protons as neutrons. Which of the
following must be true?
A) The atomic weight must be a whole number.
B) The mass number for each atom must equal the atomic weight of the element.
C) The mass number must be exactly twice the atomic number for each atom.
D) All of these are true.
47) Three atoms have the following properties.
Proton
Neutron
Electron
Atom X
119
119
119
Atom Y
119
118
119
Atom Z
118
118
119
The elements X and Y are best described as
A) isotopes.
B) cations.
C) different elements.
D) anions.

48) Three atoms have the following properties.
Proton
Neutron
Electron
Atom X
119
119
119
Atom Y
119
118
119
Atom Z
118
118
119
The elements Y and Z are best described as
A) isotopes.
B) cations.
C) different elements.
D) anions.
49) Three atoms have the following properties.
Proton
Neutron
Electron
Atom X
119
119
119
Atom Y
119
119
118
Atom Z
118
118
119
Which of the following statements is true?
A) Element Y and Z are isotopes of X.
B) Element Y is an isotope of Z.
C) Element Y is an ion of X.
D) Element Z is an ion of Y.
50) What is the identity of the element with 6 protons, 7 neutrons, and 6 electrons?
A) C
B) N
C) Al
D) Mg

51) The smallest sample of carbon atoms that can be observed with the naked eye has a mass of
approximately 2 × 10-8 g. Given that 1 g = 6.02 × 1023 amu, and that carbon has an atomic weight of 12.01
amu, determine the number of carbon atoms present in the sample.
A) 1 × 1015
B) 1 × 1016
C) 1 × 1017
D) 6 × 1023
52) An element has two naturally occurring isotopes. One has an abundance of 37.4% and an isotopic
mass of 184.953 amu, and the other has an abundance of 62.6% and a mass of 186.956 amu. What is the
atomic weight of the element?
A) 185.702 amu
B) 185.954 amu
C) 186.207 amu
D) 186.956 amu
53) The element antimony has an atomic weight of 121.757 amu and only two naturally-occurring
isotopes. One isotope has an abundance of 57.3% and an isotopic mass of 120.904 amu. Based on these
data, what is the mass of the other isotope?
A) 121.757 amu
B) 122.393 amu
C) 122.610 amu
D) 122.902 amu

54) What is the mass of one atom of the element hydrogen?
A) 2.0 g
B) 1.0 g
C) 3.4 × 10-24 g
D) 1.7 × 10-24 g
55) How many moles and how many atoms of zinc are in a sample weighing 34.9 g?
A) 0.533 mol, 8.85 × atoms
B) 0.533 mol, 3.21 × atoms
C) 1.87 mol, 3.10 × atoms
D) 1.87 mol, 1.13 × atoms
56) Steel is galvanized by giving it a surface coating of zinc. Galvanized steel is an example of
A) a compound.
B) an element.
C) a mixture.
D) an ion.
57) How many electrons are in the ion, Zn2+?
A) 28
B) 30
C) 32
D) 65

58) How many electrons are in the ion, P3-?
A) 12
B) 18
C) 28
D) 34
59) What is the identity of element Q if the ion Q2+ contains 10 electrons?
A) C
B) O
C) Ne
D) Mg
60) How many electrons are in the ion, CO32-?
A) 16
B) 28
C) 30
D) 32
61) What type of bonding is found in the compound PCl5?
A) covalent bonding
B) hydrogen bonding
C) ionic bonding
D) metallic bonding

62) Which of the species below has 28 protons and 26 electrons?
A) Fe2+
B) Ni2+
C) Fe
D) Ni
63) Butyric acid has the structural formula given below.
What is the molecular or chemical formula for butyric acid?
A) CHO
B) C2H4O
C) C4H8O2
D) C5H8O3
64) The solid compound, Na2CO3, contains
A) Na+, C4+, and O2- ions.
B) Na+ ions and CO32-ions.
C) Na2+ and CO32- ions.
D) Na2CO3 molecules.

65) Which of the following statements concerning ionic compounds is true?
A) Essentially all ionic compounds are solids at room temperature and pressure.
B) Ionic compounds do not contain any covalent bonds.
C) Ionic compounds contain the same number of positive ions as negative ions.
D) The chemical formula for an ionic compound must show a nonzero net charge.
66) The gas Freon-11, CCl3F, contains
A) C4+, Cl-, and F- ions.
B) C4+, Cl3-, and F- ions.
C) C4+ and Cl3F4- ions.
D) CCl3F molecules.
67) The definitive distinction between ionic bonding and covalent bonding is that
A) ionic bonding involves a sharing of electrons and covalent bonding involves a transfer of electrons.
B) ionic bonding involves a transfer of electrons and covalent bonding involves a sharing of electrons.
C) ionic bonding requires two nonmetals and covalent bonding requires a metal and a nonmetal.
D) covalent bonding requires two nonmetals and ionic bonding requires a metal and a nonmetal.
68) The formula for dinitrogen trioxide is
A) N(OH)3.
B) (NO3)2.
C) N2O3.
D) N3O2.

69) The chemical formula for potassium peroxide is
A) KOH.
B) KO2.
C) K2O.
D) K2O2.
70) By analogy with the oxoanions of sulfur, H2TeO3 would be named
A) hydrotellurous acid.
B) pertelluric acid.
C) telluric acid.
D) tellurous acid.
71) The ions ClO4-, ClO3-, ClO2-, and ClO- are named respectively
A) hypochlorate, chlorate, chlorite, perchlorite.
B) hypochlorite, chlorite, chlorate, perchlorate.
C) perchlorate, chlorate, chlorite, hypochlorite.
D) perchlorite, chlorite, chlorate, hypochlorate.
72) The compound, NO2, is named
A) nitrate.
B) nitrite.
C) nitrogen dioxide.
D) nitrogen(IV) oxide.

73) The ion NO2- is named
A) nitrate ion.
B) nitrite ion.
C) nitrogen dioxide ion.
D) nitrogen(II) oxide ion.
74) The thiosulfate ion is
A) HS-.
B) HSO42-.
C) SO52-.
D) S2O32-.
75) KH2PO4 is
A) hydropotassium phosphate.
B) potassium dihydrogen phosphate.
C) potassium diphosphate.
D) potassium hydrogen(II) phosphate.
76) What is the name of the compound formed between Ca and N?
A) calcium dinitride
B) calcium trinitride
C) monocalcium trinitride
D) calcium nitride

77) How many of the following names are correct?
PCl5
potassium pentachloride
NaCN
sodium cyanide
KrF4
krypton tetrafluoride
Fe(NO3)2
iron (II) nitrate
A) 1
B) 2
C) 3
D) 4
78) What group of elements does the shaded area in the following periodic table indicate?
A) alkali metals
B) alkaline earth metals
C) inner transition metals
D) transition metals

79) What group of elements does the shaded area in the following periodic table indicate?
A) alkali metals
B) alkaline earth metals
C) inner transition metals
D) transition metals
80) What group of elements does the shaded area in the following periodic table indicate?
A) alkali metals
B) alkaline earth metals
C) halogens
D) noble gases

81) What group of elements does the shaded area in the following periodic table indicate?
A) alkali metals
B) alkaline earth metals
C) halogens
D) noble gases

82) What group of elements does the shaded area in the following periodic table indicate?
A) alkali metals
B) alkaline earth metals
C) halogens
D) noble gases

83) What group of elements does the shaded area in the following periodic table indicate?
A) alkali metals
B) alkaline earth metals
C) halogens
D) noble gases

84) What group of elements does the shaded area in the following periodic table indicate?
A) gases
B) metals
C) nonmetals
D) semimetals

85) What group of elements does the shaded area in the following periodic table indicate?
A) gases
B) metals
C) nonmetals
D) semimetals

86) What group of elements does the shaded area in the following periodic table indicate?
A) gases
B) metals
C) nonmetals
D) semimetals
87) Which element is most chemically similar to the element indicated by the letter E in the following
periodic table?
A) A
B) B
C) C
D) D

88) Which element is most chemically similar to the element indicated by the letter E in the following
periodic table?
A) A
B) B
C) C
D) D
89) Which element is most chemically similar to the element indicated by the letter E in the following
periodic table?
A) A
B) B
C) C
D) D

90) Which element indicated by letter in the following periodic table reacts rapidly with water to form an
alkaline solution?
A) A
B) B
C) C
D) D
91) Which element indicated by letter in the following periodic table reacts rapidly with water to form an
alkaline solution?
A) A
B) B
C) C
D) D

92) Which element indicated by letter in the following periodic table reacts rapidly with water to form an
alkaline solution?
A) A
B) B
C) C
D) D
93) Which element indicated by letter in the following periodic table is a gas at room temperature and a
pressure of 1.0 atm?
A) A
B) B
C) C
D) D

94) Which element indicated by letter in the following periodic table is a gas at room temperature and a
pressure of 1.0 atm?
A) A
B) B
C) C
D) D
95) Which element indicated by letter in the following periodic table is a gas at room temperature and a
pressure of 1.0 atm?
A) A
B) B
C) C
D) D

96) Which element indicated by letter in the following periodic table is the poorest conductor of electricity
and heat?
A) A
B) B
C) C
D) D
97) Which element indicated by letter in the following periodic table is the best conductor of electricity
and heat?
A) A
B) B
C) C
D) D

Use the periodic table below to answer the following questions.
98) Which elements commonly form anions?
A) A and B
B) A and C
C) B and D
D) C and D
99) Which elements commonly form cations?
A) A and B
B) A and C
C) B and D
D) C and D
100) Which elements commonly form covalent bonds?
A) A and B
B) A and C
C) B and D
D) C and D

Use the periodic table below to answer the following questions.
101) Which is the correct formula of the binary fluoride of element A?
A) AF2
B) AF3
C) AF5
D) AF6
102) Which is the correct formula of the binary fluoride of element B?
A) BF2
B) BF3
C) BF5
D) BF6
103) In which pair are both formulas of binary fluorides of element C correct?
A) CF2 and CF3
B) CF2 and CF6
C) CF3 and CF5
D) CF5 and CF6

104) In which pair are both formulas of binary fluorides of element D correct?
A) DF2 and DF3
B) DF2 and DF6
C) DF3 and DF5
D) DF5 and DF6
105) Which is most likely to form a binary oxide with the formula MO (where M = element A, B, C, or D)?
A) element A
B) element B
C) element C
D) element D
106) Which is most likely to form a binary oxide with the formula MO3 (where M = element A, B, C, or
D)?
A) element A
B) element B
C) element C
D) element D
107) Which is most likely to form a binary oxide with the formula M2O3 (where M = element A, B, C, or
D)?
A) element A
B) element B
C) element C
D) element D

108) Which is most likely to form a binary oxide with the formula M4O10 (where M = element A, B, C, or
D)?
A) element A
B) element B
C) element C
D) element D
109) Assume that the mixture of substances in drawing (1) undergoes a chemical reaction. Which of the
drawings (2)-(4) represents a product mixture that is consistent with the law of mass conservation?
A) drawing (2)
B) drawing (3)
C) drawing (4)

110) Assume that the mixture of substances in drawing (1) undergoes a chemical reaction. Which of the
drawings (2)-(4) represents a product mixture that is consistent with the law of mass conservation?
A) drawing (2)
B) drawing (3)
C) drawing (4)
111) Which of the following drawings depicts a chemical reaction consistent with Dalton's atomic theory?
A) drawing a)
B) drawing b)
C) drawing c)
D) drawing d)

112) Which of the following drawings depicts a chemical reaction consistent with Dalton's atomic theory?
A) drawing a)
B) drawing b)
C) drawing c)
D) drawing d)
113) If shaded and unshaded spheres represent atoms of different elements, as shown in drawing (1),
which drawings (2)-(4) represent the law of multiple proportions?
A) only drawings (2) and (3)
B) only drawings (2) and (4)
C) only drawings (3) and (4)
D) drawings (2), (3), and (4)

114) If shaded and unshaded spheres represent atoms of different elements, as shown in drawing (1),
which drawings (2)-(4) represent the law of multiple proportions?
A) only drawings (2) and (3)
B) only drawings (2) and (4)
C) only drawings (3) and (4)
D) drawings (2), (3), and (4)
115) If shaded and unshaded spheres represent atoms of different elements, as shown in drawing (1),
which drawings (2)-(4) represent the law of multiple proportions?
A) only drawings (2) and (3)
B) only drawings (2) and (4)
C) only drawings (3) and (4)
D) drawings (2), (3), and (4)

116) If shaded and unshaded spheres represent atoms of different elements, as shown in drawing (1),
which drawings (2)-(4) represent the law of multiple proportions?
A) only drawings (2) and (3)
B) only drawings (2) and (4)
C) only drawings (3) and (4)
D) drawings (2), (3), and (4)
117) If shaded and unshaded spheres represent atoms of different elements, as shown in drawing (1),
which combination of drawings (2)-(4) represent the law of multiple proportions?
A) only drawings (2) and (3)
B) only drawings (2) and (4)
C) only drawings (3) and (4)
D) drawings (2), (3), and (4)

118) Which of the above drawings represents a pure element?
A) drawing (a)
B) drawing (b)
C) drawing (c)
119) Which of the above drawings represents a pure compound?
A) drawing (a)
B) drawing (b)
C) drawing (c)
120) Which of the above drawings represents a mixture?
A) drawing (a)
B) drawing (b)
C) drawing (c)

121) Which of the following drawings represents a collection of acetylene (C2H2) molecules? The shaded
spheres represent carbon atoms and the unshaded spheres represent hydrogen atoms.
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
122) If unshaded spheres represent sulfur atoms and shaded spheres represent oxygen atoms, which of
the following drawings depicts a collection of sulfur trioxide molecules?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)

123) Which of the above drawings represents an Ar atom?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
124) Which of the above drawings represents a Cl- ion?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
125) Which of the above drawings represents a Ca2+ ion?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
126) Which of the above drawings represents a K+ ion?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)

127) Which of the following figures represents H? Unshaded spheres represent neutrons and shaded
spheres represent protons.
A) figure (1)
B) figure (2)
C) figure (3)
D) figure (4)
128) Which of the following figures represents B? Unshaded spheres represent neutrons and shaded
spheres represent protons.
A) figure (1)
B) figure (2)
C) figure (3)
D) figure (4)
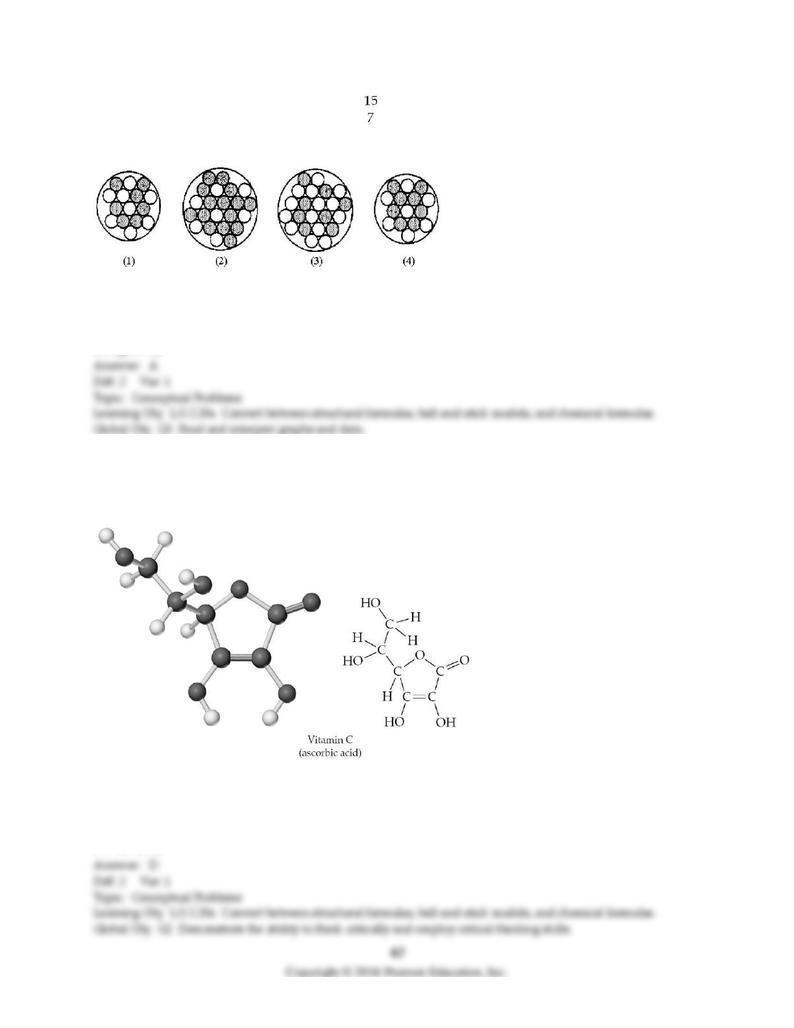
129) Which of the following figures represents N? Unshaded spheres represent neutrons and shaded
spheres represent protons.
A) figure (1)
B) figure (2)
C) figure (3)
D) figure (4)
130) Give the molecular formula corresponding to the following ball-and-stick molecular representation
of vitamin C (ascorbic acid) (gray = C, unshaded = H, black = O). In writing the formula, list the atoms in
alphabetical order.
A) CHO
B) C3H4O3
C) C6H4O6
D) C6H8O6
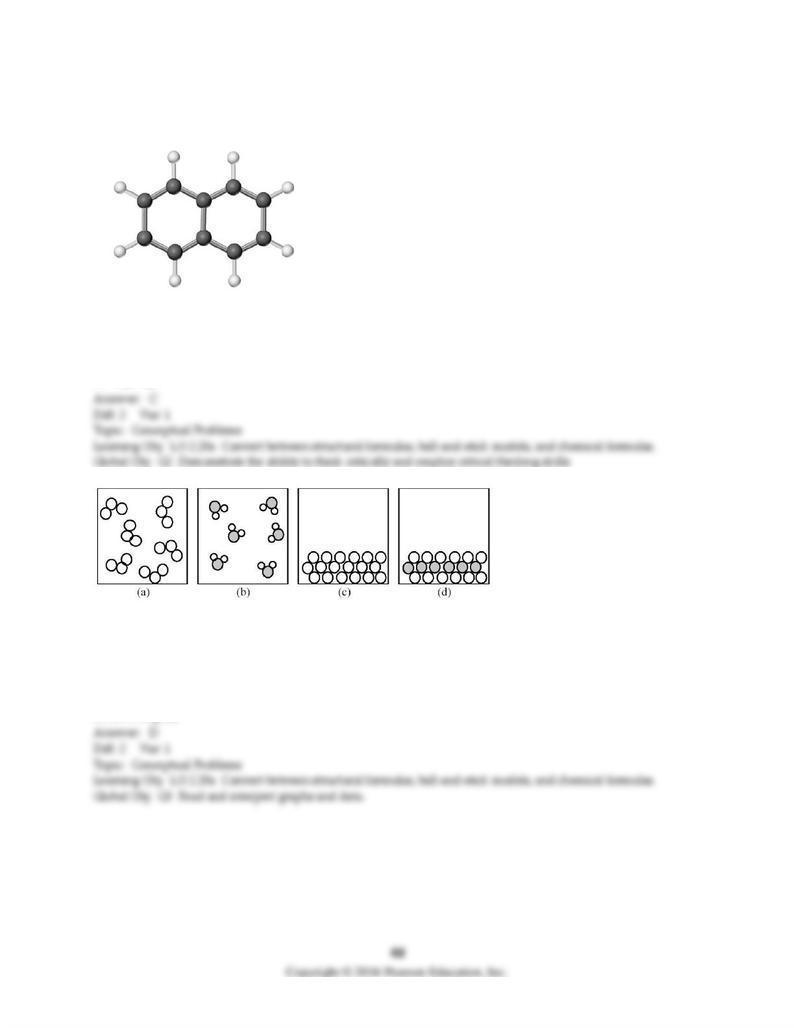
131) Give the molecular formula corresponding to the following ball-and-stick molecular representation
of naphthalene (gray = C, unshaded = H). In writing the formula, list the atoms in alphabetical order.
A) CH
B) C5H4
C) C10H8
D) C10H10
132) If shaded and unshaded spheres represent atoms of different elements, which of the above drawings
most likely represents an ionic compound at room temperature and a pressure of 1 atm?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)

133) If shaded and unshaded spheres represent atoms of different elements, which of the above drawings
most likely represents a molecular compound at room temperature and a pressure of 1 atm?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
134) If shaded and unshaded spheres represent atoms of different elements, which of the above drawings
most likely represents an ionic compound at room temperature and a pressure of 1 atm?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
135) If shaded and unshaded spheres represent atoms of different elements, which of the above drawings
most likely represents a molecular compound at room temperature and a pressure of 1 atm?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)

2.2 Algorithmic Questions
1) What is the chemical symbol for thallium?
A) Ti
B) Tl
C) Tm
D) Th
2) What is the chemical symbol for arsenic?
A) Ac
B) Ar
C) As
D) At
3) What is the chemical symbol for niobium?
A) Au
B) Nb
C) Pb
D) Nn
4) What is the chemical symbol for platinum?
A) Pd
B) Pr
C) Pt
D) Au

5) Which element has the chemical symbol, Au?
A) antimony
B) americium
C) gold
D) lead
6) What is the chemical symbol for carbon?
A) Co
B) Cr
C) C
D) Ca
7) Which element has the chemical symbol, Pb?
A) tin
B) lead
C) mercury
D) potassium
8) Which element has the chemical symbol, N?
A) nickel
B) niobium
C) nitrogen
D) nobelium

9) Which of the following elements has chemical properties similar to oxygen?
A) neon
B) hydrogen
C) nitrogen
D) tellerium
10) ________ is used in lights and signs.
A) Neon
B) Helium
C) Iodine
D) Silicon
11) ________ does not combine with any other element.
A) Chlorine
B) Nitrogen
C) Helium
D) Krypton
12) Identify a chemical property.
A) tarnishing
B) boiling point
C) taste
D) solubility

13) ________ is a nonmetal that is a solid at room temperature.
A) Calcium
B) Selenium
C) Bromine
D) Copper
14) Rubidium belongs to the ________ group of the periodic table.
A) alkali metal
B) alkaline earth metal
C) halogen
D) noble gas
15) Chlorine belongs to the ________ group of the periodic table.
A) alkali metal
B) alkaline earth metal
C) halogen
D) noble gas
16) Radon belongs to the ________ group of the periodic table.
A) alkali metal
B) alkaline earth metal
C) halogen
D) noble gas

17) Calcium belongs to the ________ group of the periodic table.
A) alkali metal
B) alkaline earth metal
C) halogen
D) noble gas
18) Which of the following elements has chemical properties similar to oxygen?
A) fluorine
B) hydrogen
C) nitrogen
D) sulfur
19) Which of the following elements is a gas at room temperature?
A) bromine
B) iron
C) krypton
D) sodium
20) Which of the following elements is not a solid at room temperature?
A) Ag
B) Al
C) He
D) Fe

21) Which of the following elements is classified as a semimetal?
A) calcium
B) silicon
C) fluorine
D) uranium
22) Which of the following elements is a good conductor of heat and electricity?
A) carbon
B) chlorine
C) neon
D) zinc
23) Which one of the following elements is a poor conductor of heat and electricity?
A) copper
B) phosphorus
C) iron
D) lead
24) All of the following elements are nonmetals except
A) beryllium.
B) carbon.
C) hydrogen.
D) oxygen.

25) Which of the following underlined items is not an intensive property?
A) the amount of gold.
B) the color of copper hydroxide
C) the density of argon
D) the melting point of iron metal
26) Which of the following underlined items is not an extensive property?
A) the color of a cobalt compound
B) the diameter of a gold nugget
C) the mass of a diamond
D) the volume of a glucose solution
27) Which group 1A element is not a metal?
A) H
B) K
C) Cs
D) Be
28) Which of the following elements is a liquid at room temperature?
A) neon
B) helium
C) mercury
D) lithium

29) Which of the following elements is not a solid at room temperature?
A) Zn
B) Hg
C) N
D) C
30) Which of the following elements is classified as a semimetal?
A) gold
B) astatine
C) osmium
D) berkelium
31) Which of the following elements is a good conductor of heat and electricity?
A) silicon
B) iodine
C) radon
D) lead
32) Which one of the following elements is a poor conductor of heat and electricity?
A) nickel
B) sulfur
C) aluminum
D) lead

33) All of the following elements are nonmetals except
A) copper.
B) nitrogen.
C) krypton.
D) phosphorus.
34) Methane and oxygen react to form carbon dioxide and water. What mass of water is formed if 6.4 g of
methane reacts with 25.6 g of oxygen to produce 17.6 g of carbon dioxide?
A) 14.4 g
B) 17.6 g
C) 29.6 g
D) 32.0 g
35) Sodium metal and water react to form hydrogen and sodium hydroxide. If 11.96 g of sodium react
with water to form 0.52 g of hydrogen and 20.80 g of sodium hydroxide, what mass of water was
involved in the reaction?
A) 9.36 g
B) 11.96 g
C) 20.28 g
D) 21.32 g

36) A sample of pure lithium chloride contains 16% lithium by mass. What is the % lithium by mass in a
sample of pure lithium carbonate that has twice the mass of the first sample?
A) 8.20%
B) 16.4%
C) 32.8%
D) 65.6%
37) A sample of pure calcium fluoride with a mass of 15.0 g contains 7.70 g of calcium. How much
calcium is contained in 35.0 g of calcium fluoride?
A) 1.99 g
B) 7.70 g
C) 15.0 g
D) 18.0 g
38) Mg can react with HCl to produce the white solid MgCl2 and H2 gas. A student mixes 1.99 g of Mg
with 5.98 g of HCl. If the mass of the white solid is 7.79 g, then what is the mass of H2 produced?
A) 0.0 g
B) 0.18 g
C) 2.0 g
D) 15.76 g

39) Elements A and Q form two compounds, AQ and A2Q3. The mass ratio (mass Q)/(mass A) for AQ is
0.286. What is the mass ratio (mass Q)/(mass A) for A2Q3?
A) 0.191
B) 0.429
C) 2.33
D) 5.24
40) A proton is approximately
A) 600 times larger than an electron.
B) 2000 times larger than an electron.
C) 700 times smaller than an electron.
D) 3000 times smaller than an electron.
41) Which are isotopes? An atom that has an atomic number of 12 and a mass number of 26 is an isotope
of an atom that has
A) an atomic number of 13 and a mass number of 26.
B) an atomic number of 12 and a mass number of 24.
C) 12 neutrons and 14 protons.
D) 12 protons and 14 neutrons.
42) Which of the following represent isotopes?
A: [ ] B: [ ] C: [ ] D: [ ]
A) A and B
B) A and C
C) A and D
D) C and D

43) How many protons (p) and neutrons (n) are in an atom of Ra?
A) 88 p, 138 n
B) 88 p, 226 n
C) 138 p, 88 n
D) 226 p, 88 n
44) How many protons (p) and neutrons (n) are in an atom of calcium-46?
A) 20 p, 26 n
B) 20 p, 46 n
C) 26 p, 20 n
D) 46 p, 20 n
45) What is the element symbol for an atom that has 33 protons and 41 neutrons?
A) As
B) Nb
C) O
D) W
46) How many electrons are in a neutral atom of iodine-131?
A) 1
B) 53
C) 54
D) 131

47) Identify the chemical symbol of element Q in Q.
A) As
B) Mo
C) Os
D) Se
48) Which are isotopes? An atom that has an atomic number of 35 and a mass number of 76 is an isotope
of an atom that has
A) an atomic number of 31 and a mass number of 76.
B) an atomic number of 35 and a mass number of 80.
C) 41 neutrons and 35 protons.
D) 41 protons and 35 neutrons.
49) The isotope represented by C is named
A) carbon-6.
B) carbon-3.
C) carbon-9.
D) carbon-15.
50) A bottle of pure element was missing part of a label. The label said 2.258 x 1023 atoms. You
determine the mass of the elements in the bottle to be 10.51946. What is the identity of this element?
A) B
B) N
C) Si
D) Sr

51) What is the standard isotope that is used to define the number of atoms in a mole?
A) 14N
B) 12C
C) 9Be
D) 31P
52) The number of atoms of carbon in 28 g of silicon is
A) 28
B) 28 × 6.022 × 1022
C) 2.8 × 1023
D) 6.022 × 1023
53) One mole of which element has the smallest mass?
A) Co
B) Zn
C) Ni
D) Ru
54) 24.0 g of which element contains the greatest number of atoms?
A) Be
B) C
C) O
D) Na

55) A banana split is an example of
A) a compound.
B) an element.
C) a mixture.
D) an ion.
56) Hydrochloric acid is an example of
A) a compound.
B) an element.
C) an ion.
D) a mixture.
57) Iodine is an example of
A) a compound.
B) an element.
C) a mixture.
D) an ion.
58) Water is an example of
A) a compound.
B) an element.
C) a mixture.
D) an ion.

59) A cake is an example of
A) a compound.
B) an element.
C) mixture.
D) an an anion.
60) Orange juice is an example of
A) a compound.
B) an element.
C) an ion.
D) a mixture.
61) Aluminum is an example of
A) a compound.
B) an element.
C) a mixture.
D) an ion.
62) Ethane is an example of
A) a compound.
B) an element.
C) a mixture.
D) a cation.

63) In which set do all elements tend to form cations in binary ionic compounds?
A) Li, B, O
B) Mg, Cr, Pb
C) N, As, Bi
D) O, F, Cl
64) How many electrons are in the ion, Fe2+?
A) 24
B) 26
C) 28
D) 56
65) How many electrons are in the ion, S2-?
A) 14
B) 18
C) 30
D) 34
66) In which of the following sets do all species have the same number of electrons?
A) Cl-, Ar, Ca2+
B) N, O2-, F-
C) Sc3+, Y3+, La3+
D) Cr, Cr2+, Cr3+

67) In which of the following sets do all species have the same number of protons?
A) At-, Rn, Ra2+
B) C, N3-, O2-
C) CO3+, Rh3+, Ir3+
D) Br, Co2+, Co3+
68) In which of the following sets do all species have the same number of electrons?
A) I-, Xe, Cs2+
B) C, N3-, O2-
C) Mg2+, Ca2+, Ba2+
D) S, S2-, S2+
69) In which of the following sets do all species have the same number of protons?
A) Br-, Kr, Rb2+
B) C, N3-, O2-
C) Mg2+, Ca2+, Ba2+
D) O, O2-, O2+
70) What is the identity of element Q if the ion Q2+ contains 18 electrons?
A) Si
B) S
C) Ar
D) Ca

71) How many electrons are in the ion, SO42-?
A) 26
B) 46
C) 48
D) 50
72) In which set do all elements tend to form anions in binary ionic compounds?
A) Cs, B, O
B) Ca, Zn, Pb
C) N, Sb, Bi
D) S, Cl, Br
73) What type of bonding is found in the compound NH3?
A) covalent bonding
B) hydrogen bonding
C) ionic bonding
D) metallic bonding
74) Which one of the following compounds contains ionic bonds?
A) MgO
B) HCl
C) PCl3
D) CO2

75) Which of the following is the correct chemical formula for a molecule of iodine?
A) I
B) I-
C) I+
D) I2
76) Which of the compounds, Ca H2, H2O , C H4, XeF4 are ionic compounds?
A) only C H4
B) only Ca H2
C) Ca H2 and Xe F4
D) H2O , C H4, and XeF4
77) Which of the compounds C2H6, CaCl2, Cu(NO3)2, OF2 are expected to exist as molecules?
A) only C2H6
B) C2H6 and OF2
C) C2H6, Cu(NO3)2, and OF2
D) CaCl2 and Cu(NO3)2
78) Which of the following elements has the least tendency to form an ion?
A) Be
B) H
C) He
D) O

79) The solid compound, Na4SiO4, contains
A) Na+, Si4+, and O2- ions.
B) Na+ ions and SiO44- ions.
C) Na4+ and SiO44- ions.
D) Na4SiO4 molecules.
80) What is the chemical formula for nickel(II) phosphate?
A) Ni2P
B) Ni2PO4
C) Ni3P2
D) Ni3(PO4)2
81) What is the charge on the Sc ions in Sc2O3?
A) 2-
B) 1+
C) 2+
D) 3+
82) Na2S is named
A) sodium disulfide.
B) sodium sulfide.
C) sodium(II) sulfide.
D) sodium sulfur.

83) What is the chemical formula for strontium hydroxide?
A) SrH2
B) SrOH
C) SrOH2
D) Sr(OH)2
84) What is the chemical formula for radium hydride?
A) RaH2
B) RaOH
C) RaOH2
D) Ra(OH)2
85) An aqueous solution of H Cl is named
A) hydrochloric acid.
B) hydrochlorous acid.
C) chloric acid.
D) chlorous acid.
86) The chemical formula for the nitrite ion is
A) N2-.
B) N 3-.
C) NO2-.
D) NO3-.

87) The chemical formula for rubidium peroxide is
A) RbOH.
B) RbO2.
C) Rb2O.
D) Rb2O2.
88) The compound, Cu(I O3 )2, is named
A) copper iodate(II).
B) copper(I) iodate.
C) copper(I) iodate(II).
D) copper(II) iodate.
89) The compound, ClO , is named
A) chlorite.
B) hypochlorite.
C) chlorine monoxide.
D) chlorine (II) oxide.
90) The ion, NO2-, is named
A) nitrate ion.
B) nitrite ion.
C) nitrogen dioxide ion.
D) nitrogen(II) oxide ion.

91) The chemical formula for chlorous acid is
A) H ClO(aq).
B) H ClO2(aq).
C) H ClO3(aq).
D) H Cl O4(aq).
92) The chemical formula for magnesium nitride is
A) Mg(NO3)2.
B) Mg(NO2)2.
C) Mg3N2.
D) MgN2.
93) In which set do all elements tend to form cations in binary ionic compounds?
A) Na, B, S
B) Ca, Cr, Pb
C) S, As, Bi
D) O, Br, I
94) In which set do all elements tend to form anions in binary ionic compounds?
A) C, S, Pb
B) K, Fe, F
C) Na, Ba, Al
D) N, O, Cl

95) What is the most likely charge on an ion of phosphorus, P?
A) 5-
B) 3-
C) 1+
D) 5+
96) Which element can form more than one kind of monatomic ion?
A) Sr
B) Al
C) Sn
D) O
97) Which element can form more than one kind of monatomic ion?
A) Na
B) I
C) Cr
D) Zn
98) Which one of the following compounds contains ionic bonds?
A) MgS
B) HF
C) NCl3
D) SiO2

99) Which of the following is the correct chemical formula for a molecule of nitrogen?
A) N
B) N-
C) N+
D) N2
100) Which of the compounds, Na3P, PH3, C2H6, IBr3, are ionic compounds?
A) only C2H6
B) only Na3P
C) Na3P and PH3
D) PH3, C2H6, and IBr3
101) Which of the compounds, C5H12, CaF2, Pd(NO3)2, OCl2, are expected to exist as molecules?
A) only C5H12
B) C5H12 and OCl2
C) C5H12, Pd(NO3)2, and OCl2
D) CaF2 and Pd(NO3)2
102) Which of the following elements has the least tendency to form an ion?
A) Ca
B) Li
C) Kr
D) S

103) What is the chemical formula for iron(II) phosphate?
A) Fe2P
B) Fe2PO4
C) Fe3P4
D) Fe3(PO4)2
104) What is the charge on the In in the ionic compound In2Te3?
A) 2-
B) 1+
C) 2+
D) 3+
105) Na2O is named
A) sodium dioxide.
B) sodium oxide.
C) sodium(II) oxide.
D) sodium oxygen.
106) What is the chemical formula for cesium bicarbonate?
A) Cs2HCO3
B) CsHCO
C) CsHCO2
D) CsHCO3

107) What is the chemical formula for calcium chromate?
A) CaCrO2
B) CaCrO
C) CaCrO3
D) CaCrO4
108) The chemical formula for the carbonate ion is
A) C-.
B) CO-.
C) CO32-.
D) CO22-.
109) The compound, Sn(IO3)2, is named
A) tin iodate(II).
B) tin(I) iodate.
C) tin(I) iodate(II).
D) tin(II) iodate.
110) The chemical formula for calcium telluride is
A) Ca(TeO3).
B) Ca(TeO2).
C) CaTe.
D) CaTe2.

111) What are the names of the ions Mn2+, Sn2+, and Se2-?
A) manganese, tin, and selenium
B) manganese, tin(II), and selenide
C) manganese(II), tin(II), and selenium(II-)
D) manganous, stannous, and selenide
112) Which drawing represents the ionic compound Sr3(PO4)2?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
113) Which drawing represents the ionic compound Ag2CO3?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)

114) Which drawing represents the ionic compound BaF2?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
115) Which drawing represents the ionic compound AgClO3?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
116) Which drawing represents the ionic compound RbClO4?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
2.3 Short Answer Questions
1) The symbol for mercury is ________.
2) Pb is the symbol for the element ________.

3) In a periodic table rows are called ________ and columns are called ________.
4) The element Al can be found in period ________ and group ________ of the periodic table.
5) The element that is in period 5 and group 2A has the symbol ________.
6) A property that depends on the amount of a substance is an ________ property, whereas a property
that is independent on the amount of substance is an ________ property.
7) Elements are classified as metals, nonmetals, or semimetals. At room temperature a certain element
exists as a dull yellow solid that is a poor conductor of electricity and is brittle. This element is most likely
a ________.
8) Sodium is an example of an ________ metal that reacts with water to form hydrogen gas and an
________ solution.

9) 81 g of HBr react with 40 g of NaOH to produce 18 g of H2O, then the number of grams of NaBr
produced is ________.
HBr + NaOH → H2O + NaBr
10) According to the law of multiple proportions, if 12 g of carbon combine with 16 g of oxygen to form
CO, the number of grams of carbon that combine with 16 g of oxygen in the formation of CO2 is
________.
11) The charge to mass ratio of an electron was determined from Rutherford's cathode-ray tube
experiment to be 1.759 × 108 C/g and the charge on a single electron was determined from the Millikan oil
drop experiment to be 1.602 × 10-19 C, so the mass of a single electron is ________.
12) The subatomic particles contained in the nucleus of an atom are ________ and ________.
13) Atoms of the same element always have the same number of ________ in their nuclei.

14) Isotopes have the same number of ________ but different numbers of ________ in their nuclei.
15) The symbol of the isotope having Z = 88 and A = 226 is ________.
16) The symbol for technetium-98 is ________.
17) The number of neutrons in a neutral atom of uranium-238 is ________.
18) A neutral atom with atomic number 5 and mass number 11 contains ________ electrons.
19) Chlorine has two common isotopes, chlorine-35 and chlorine-37, and an atomic mass of 35.45 amu.
The natural abundance of chlorine-35 is ________ (greater than, less than, the same as) the natural
abundance of chlorine-37.

20) The number of atoms in 1 g of H is ________ (greater than, less than, the same as) the number of
atoms in 12 g of C.
21) To the nearest whole number, the number of grams of Ba in 3.25 mol of Ba is ________.
22) The number of moles of Li in 34.7 g Li is ________.
23) 10% saline solution (sodium chloride dissolved in water) is an example of a ________.
24) The number of electrons in the ion Ca2+ is ________.
25) The number of electrons in the ion C4- is ________.

26) The bonding in NaI is ________, whereas the bonding in NH3 is ________.
27) Phosphate ion has the formula ________.
28) The formula of thallium(III) selenide contains ________ thallium(III) and ________ selenide ions.