
Chemistry, 7e (McMurry/Fay)
Chapter 23 Connections to Organic and Biological Chemistry
23.1 Multiple-Choice Questions
1) The most important characteristic of carbon atoms for forming organic molecules is
A) ability to bond together to form long chains.
B) ability to form multiple covalent bonds.
C) use of hybrid orbitals.
D) A and B
2) Which of the following statements about properties of organic compounds is not correct?
A) Most organic compounds are not soluble in water.
B) Most organic compounds do not conduct electricity.
C) Organic compounds have higher melting and boiling points than ionic compounds.
D) Organic compounds have weak intermolecular forces.
3) Another term for alkanes is
A) alkenes.
B) alkynes.
C) saturated hydrocarbons.
D) unsaturated hydrocarbons.
4) Compounds that have the same formula but different molecular structures are called
A) isoelectronic.
B) isomers.
C) isotones.
D) isotopes.

5) Which of the following statements about isomers is false?
A) All alkanes have branched chain isomers.
B) As the number of carbon atoms increase in a compound, so do the number of possible isomers.
C) Isomers have different physical properties.
D) Isomers have the same formula but different molecular structures.
6) How many isomers are there for C5H12?
A) 3
B) 4
C) 5
D) 6
7) Which is the condensed structure of a straight-chain hydrocarbon?
A) I
B) II
C) III
D) IV

8) Which condensed structure below representing an alkane is not correct?
A) CH3CH2CH3
B)
C) CH3CH2CH2CH3
D)
9) Which of the following condensed structures represent the same molecule?
A) I and II
B) II and IV
C) III and IV
D) II and III

10) Which of the structures below are identical?
A) I and II
B) II and III
C) III and IV
D) I and IV
11) Which one of the structures below is different from the other three?
A) I
B) II
C) III
D) IV

12) Some of the products from reaction of bromine and propane are shown below. (The hydrogen atoms
are not shown in the structures.) How many are identical structures?
A) 2
B) 3
C) 4
D) All are isomers of each other.
13) When alkanes react with chlorine in the presence of ultraviolet light, chlorine atoms substitute for one
or more alkane hydrogen atoms. What is the number of different chloroalkane compounds that can be
formed by the reaction of C2H6 with chlorine?
A) 3
B) 6
C) 9
D) 12
14) Which of the following statements about cycloalkanes is true?
A) The bonds in cyclopropane and cyclobutane are weaker than other cycloalkanes.
B) Cycloalkanes are also called "acyclic" compounds.
C) Cyclopropane has 109° bond angles.
D) Most cycloalkanes are planar molecules.

15) The cycloalkane represented by a square is called ________ and has the molecular formula ________.
A) cyclopropane, C3H6
B) cyclopropane, C3H8
C) cyclobutane, C4H4
D) cyclobutane, C4H8
16) What are the C–C–C bond angles in cyclopropane and cyclohexane?
A) 60° in both molecules
B) 60° in cyclopropane and 109.5° in cyclohexane
C) 60° in cyclopropane and 120° in cyclohexane
D) 109.5° in both molecules
17) Which compound is not an isomer of the other three?
A) cyclohexane
B) 1, 2-dimethylcyclobutane
C) 1, 2-dimethylcyclopropane
D) methylcyclopentane
18) Which of the following statements is false regarding functional groups?
A) The chemical properties of the functional group dictate the chemistry of the larger molecule.
B) Each functional group has a characteristic chemical behavior.
C) A functional group consists of an atom or a group of atoms that is part of a larger molecule.
D) A functional group consists of only carbon and hydrogen atoms.

19) Identify the functional group:
A) alcohol
B) amine
C) amide
D) ester
20) Identify the functional group:
A) aldehyde
B) ketone
C) ester
D) ether

21) Which functional group below represents a carboxylic acid?
A)
B)
C)
D)
22) Which functional group below represents a ketone?
A)
B)
C)
D)

23) Which functional group below represents an aldehyde?
A)
B)
C)
D)
24) The functional groups in the molecule below are
H2NCH2CO2H
A) alkane, amide, and carboxylic acid.
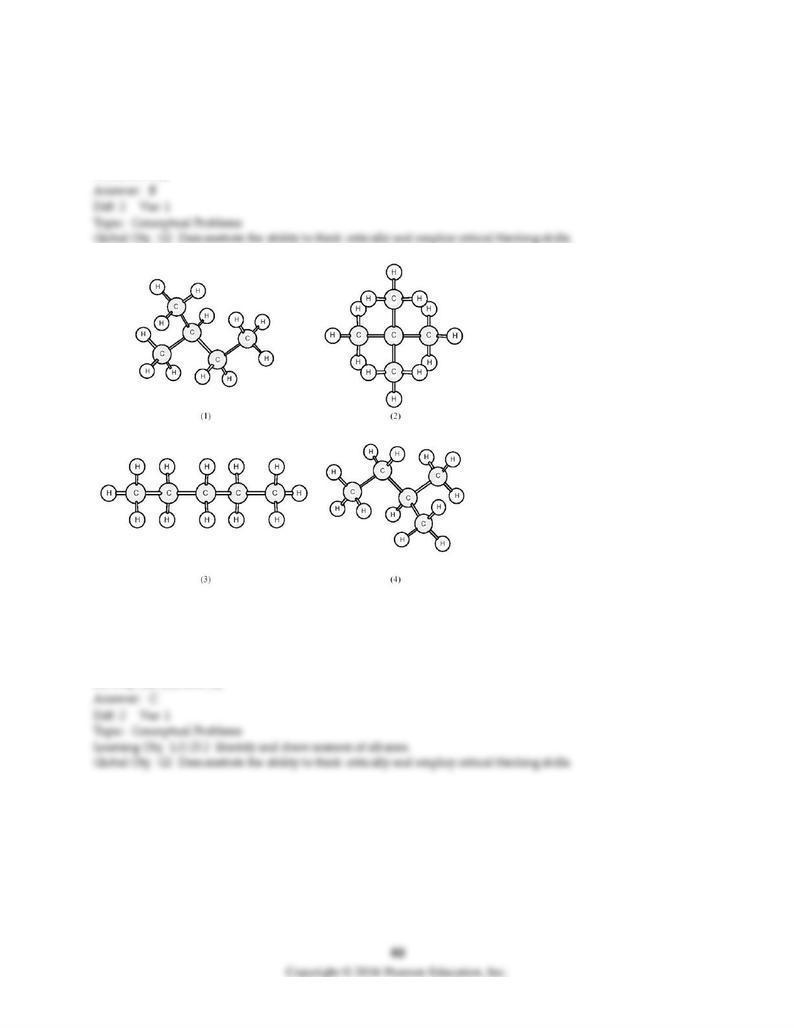
B) alkane, amine, and carboxylic acid.
C) alkane, amide, ketone, and alcohol.
D) alkane, amine, ketone, and alcohol.

25) The functional groups in the molecule below are
O O
HCCH2COCH3
A) alkane, aldehyde, and ester.
B) alkane, aldehyde, ketone, and ether.
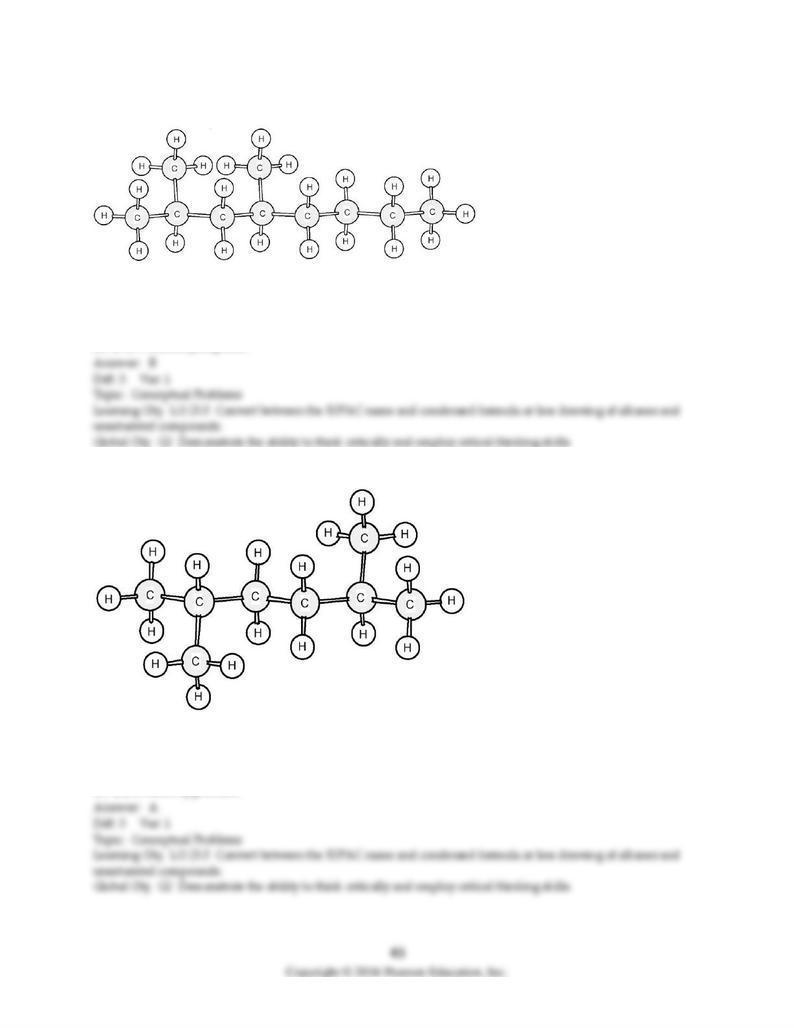
C) alkane, carboxylic acid, and ester.
D) alkane, ketone, and ester.
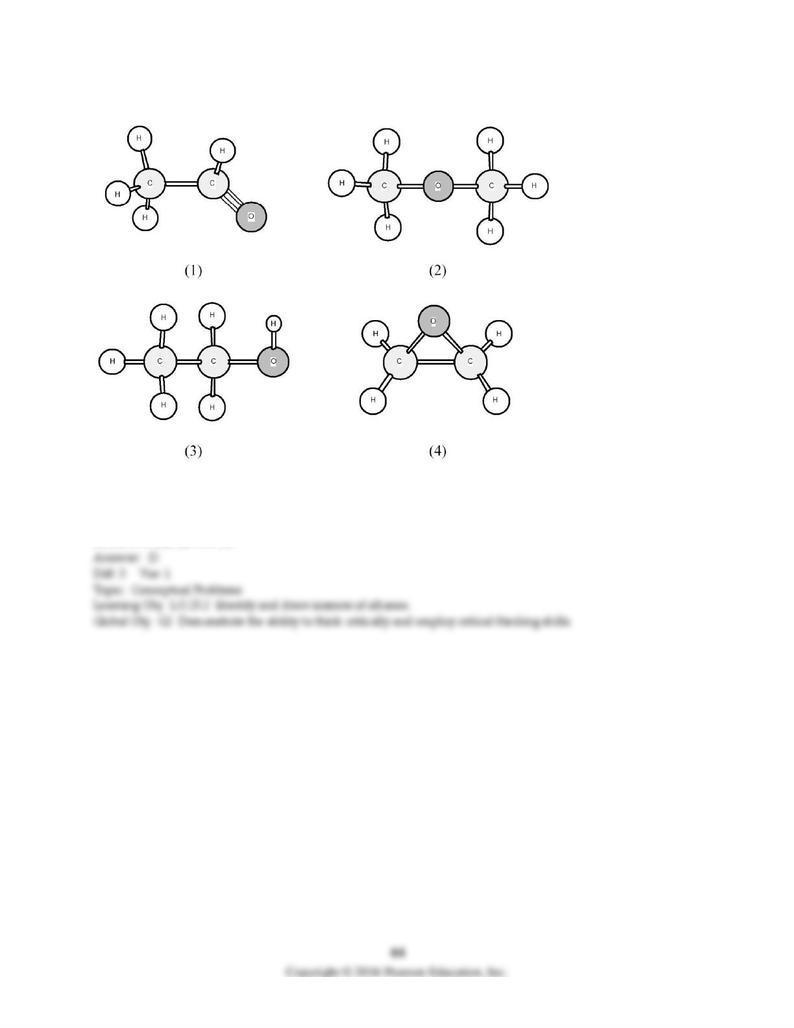
26) Which of the following compounds is most soluble in water?
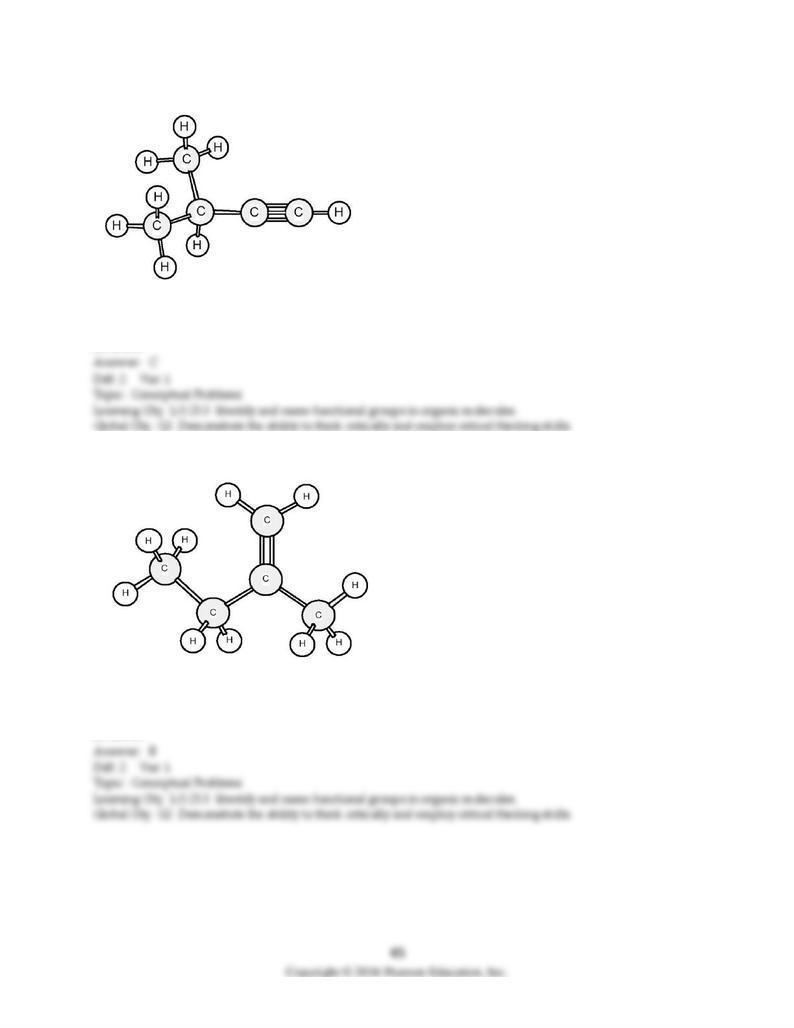
A) decanol
B) dimethyl ether
C) ethane
D) ethanol
27) How many ether isomers have the formula C4H10O?
A) 2
B) 3
C) 4
D) 5
28) What type of compound contains the -NH2 functional group?
A) alcohol
B) amine
C) carboxylic acid
D) ether

29) The C-C=O bond angle in acetic acid, shown below, is closest to
O
CH3C–OH
A) 90°.
B) 109.5°.
C) 120°.
D) 180°.
30) The H-C-H bond angles in acetic acid, shown below, are closest to
O
CH3C–OH
A) 90°.
B) 109.5°.
C) 120°.
D) 180°.
31) What is the common name for the simplest ketone, propanone?
A) acetal
B) acetone
C) carbanone
D) formalin

32) Which one of the following behaves like an acid?
A) CH3COCH3
B) (CH3)2NH
C) C2H5OH
D) C2H5COOH
33) How many grams of Ca(OH)2 are needed to neutralize 5.00 g of 3-methylbutanoic acid?
A) 1.81 g
B) 2.10 g
C) 3.63 g
D) 4.20 g
34) Which one of the following molecules is the most polar?
A) acetaldehyde
B) acetic acid
C) ethane
D) ethylene
35) Which of the functional groups below are not characterized by π bonds?
A) alkenes
B) alkynes
C) aldehydes
D) alcohols

36) Which class of hydrocarbons has the general formula CnH2n+2?
A) alkanes
B) alkenes
C) alkynes
D) cylcloalkanes
37) Which statement below is false concerning nomenclature rules for alkanes?
A) Carbon atoms are numbered starting with the atom farthest from the branching point.
B) Each branching group is assigned a number referring to its point of attachment in the parent chain.
C) The name of the alkane is written as a single word.
D) The parent name is taken from the longest continuous chain of carbon atoms.
38) An alkyl group is named for the part of the alkane group that remains after a ________ is removed.
A) C atom
B) CH3 group
C) CH2 group
D) H atom
39) If the point of attachment to an alkane is by the middle carbon in a propyl group, the group is called
A) isopropyl.
B) n-propyl.
C) sec-propyl.
D) tert-propyl.

40) What is the name for this alkyl group?
A) isobutyl
B) tert-butyl
C) tributyl
D) trimethyl
41) The following structure represents a butyl group.
A) normal
B) secondary
C) tertiary
D) quaternary

42) What is the IUPAC name for the following compound?
A) heptane
B) 3, 3-dimethylpentane
C) 2, 2-dimethyl-2-ethylpropane
D) 2, 2-diethylpropane
43) What is the IUPAC name for the following compound?
A) 1, 3-dimethyl-1-ethylhexane
B) 4, 6-dimethyl-6-ethylhexane
C) 3, 5-dimethyloctane
D) sec-hexylbutane

44) Which of the following compounds is 5-ethyl-4-isopropyl-2-methylnonane?
A)
B)
C)
D)
45) Which name is not correct?
A) 1, 2-dichloropentane
B) 2, 3-dimethylbutane
C) 2-ethylbutane
D) 4-propylheptane

46) The names of compounds with carbon-carbon double bonds contain the suffix
A) -ane.
B) -ene.
C) -yne.
D) -one.
47) What class of hydrocarbons has the general formula CnH2n?
A) alkanes
B) alkenes and cycloalkanes
C) alkynes
D) aromatics
48) What is the hybridization of the carbon in (CH3)2C*=CHC N indicated by the asterisk (*)?
A) sp
B) sp2
C) sp3
D) dsp3
49) What is the molecular geometry around the carbon in (CH3)2C=CHC* N indicated by the
asterisk (*)?
A) linear
B) trigonal planar
C) trigonal pyramidal
D) tetrahedral

50) Which of the molecules shown below are unsaturated?
(1) CH3CH2CH2CH(CH3)2
(2) CH3CH=CHCH(CH3)2
(3) HC CCH2CH(CH3)2
A) All are unsaturated.
B) Only (2) and (3) are unsaturated.
C) Only (2) is unsaturated.
D) Only (3) is unsaturated.
51) How many geometrical and structural isomers are there of butene?
A) 3
B) 4
C) 5
D) 6
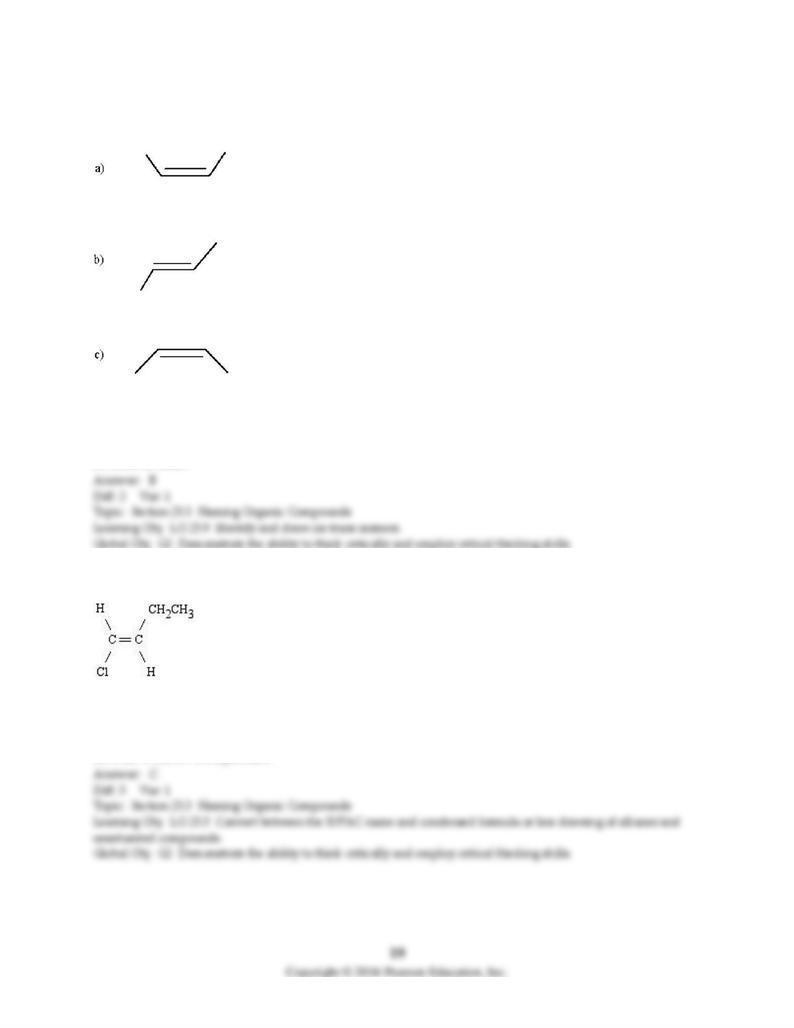
52) Which of the following is trans-2-butene? (Only bonds are shown in the skeletal structures below; the
carbon and hydrogen atoms are deleted.)
A) structure a
B) structure b
C) structure c
D) none of these
53) What is the name of the following structure?
A) cis-1-ethyl-2-chloroethane
B) cis-1-chloro-1-butene
C) trans-1-chloro-1-butene
D) trans-1-chloro-2-ethylethane

54) What is the name of the following structure?
A) tert-butylethyne
B) 3, 3-dimethyl-1-pentyne
C) 3-ethyl-3-methyl-1-butyne
D) trans-ethylmethylbutyne
55) The most important synthetic chemical reactions that alkenes and alkynes undergo are called
A) addition reactions.
B) combustion reactions.
C) photochemical halogenation reactions.
D) substitution reactions.
56) Name the product of hydrogenation of trans-2-pentene.
A) pentane
B) cis-2-pentene
C) trans-2-pentane
D) none of these

57) How many carbons are found in a molecule that contains the prefix but-?
A) 2
B) 4
C) 3
D) 7
58) "Wood alcohol" is the common name for
A) methanol.
B) ethanol.
C) 2-propanol.
D) 1, 2-ethanediol.
59) The term "carbohydrates" refers to a large class of polyhydroxylated
A) alcohols and carboxylic acids.
B) aldehydes and ketones.
C) amines and amides.
D) ethers and esters.
60) The monosaccharide shown below is a(n)
A) aldohexose.
B) aldopentose.
C) ketohexose.
D) ketopentose.

61) The monosaccharide shown below is a(n)
A) aldohexose.
B) aldopentose.
C) ketohexose.
D) ketopentose.
62) What class of compounds are cyclic?
A) amino acids
B) lipids
C) monosaccharides
D) proteins
63) In water, which form of glucose predominates?
A) cyclic α
B) cyclic β
C) open chain
D) All three are equal in solution.

64) When crystallized, which form of glucose predominates?
A) cyclic α
B) cyclic β
C) open chain
D) All three are equal in crystals.
65) Sucrose, common table sugar, when hydrolyzed will form
A) amylose and glycogen.
B) cellulose and starch.
C) glucose and fructose.
D) lactose and maltose.
66) Starch is a polymer consisting of thousands of
A) α-glucose molecules.
B) β-glucose molecules.
C) long chain fatty acids.
D) amino acids.
67) Cellulose is a polymer consisting of thousands of
A) α-glucose molecules.
B) β-glucose molecules.
C) long chain fatty acids.
D) amino acids.

68) Which substance is a monosaccharide?
A) cellulose
B) glucose
C) glycogen
D) starch
69) Which of the following statements about cis-trans isomers is not correct?
A) Conversion between cis- and trans-isomers occurs easily by rotation around the double bond.
B) In the trans- isomer, the groups of interest are on opposite sides across the double bond.
C) In the cis- isomer, the groups of interest are on the same side of the double bond.
D) There are no cis-trans isomers in alkynes.
70) Which compound will have the largest dipole moment?
A) cis-1, 2-dichloroethylene
B) trans-1, 2-dichloroethylene
C) tetrabromoethylene
D) tetrachloroethylene
71) Which compound will exhibit cis-trans isomerism?
A) 1, 2-dichloroethane
B) 1, 2-dichloroethene
C) dichloroethyne
D) ethylene

72) How many distinct isomers can be drawn for a molecule of C4H8?
A) 1
B) 2
C) 3
D) 4
73) Hydrogenation of vegetable oils converts them into what type of molecule?
A) esters
B) ethers
C) polymers
D) saturated fats
74) "Glycerol" is the common name for
A) ethanol.
B) 2-propanol.
C) 1, 2-ethanediol.
D) 1, 2, 3-propanetriol.
75) Lipids are characterized by what property?
A) chemical reactivity
B) chemical structure
C) refractive index
D) solubility

76) Which statement below regarding fatty acids is not correct? Fatty acids
A) are always liquids.
B) are long chain carboxylic acids.
C) are usually unbranched chains.
D) usually have an even number of carbon atoms.
77) All of the fatty acids below contain eighteen carbons and range from saturated to three double bonds.
Which has the highest melting point?
A) stearic acid (saturated)
B) oleic acid (one double bond)
C) linoleic acid (two double bonds)
D) linolenic acid (three double bonds)
78) What functional group is commonly found in fats?
A) —NH2
B) —OH
C) —C—O—C
D) O
—O—C—C—
79) Which of the following statements best describes the difference between vegetable oils and animal
fats?
A) Animals fats have more carboxylic acid groups.
B) Vegetable oils have carbon chains with more unsaturated bonds.
C) Vegetable oils have more alcohol groups.
D) Animal fats have more unsaturated bonds.

80) In addition to an amine, which functional group do all amino acids contain?
A) an alcohol
B) an amide
C) a carboxylic acid
D) an oxyacid
81) What does the term "alpha" amino acid mean?
A) The amino group attaches to the carbon in the carboxyl group.
B) The amino group attaches to the carbon next to the carboxylic acid group.
C) The amino acid contains only one carbon atom.
D) The amino acid contains only one carboxylic acid group.
82) What does the term "essential" mean when referring to amino acids? These are the amino acids which
A) are necessary for digestion.
B) are necessary for respiration.
C) are synthesized in our bodies.
D) we must obtain from our diet.
83) Which is an amino acid nonpolar neutral side chain?
A) -CH3
B) -CH2OH
C) -CH2CO2H
D) -CH2CH2CH2CH2NH2

84) Which is an amino acid polar neutral side chain?
A) -CH3
B) -CH2OH
C) -CH2CO2H
D) -CH2CH2CH2CH2NH2
85) Which is an amino acid basic side chain?
A) -CH3
B) -CH2OH
C) -CH2CO2H
D) -CH2CH2CH2CH2NH2
86) Which is an amino acid acidic side chain?
A) -CH3
B) -CH2OH
C) -CH2CO2H
D) -CH2CH2CH2CH2NH2
87) What does the term "hydrophobic" mean?
A) water reactive
B) water repelling
C) water soluble
D) none of these

88) Peptide bonds that link amino acids together in proteins contain the ________ functional group.
A) amine
B) amide
C) ester
D) ketone
89) In the protein, Val-Tyr-His-Pro, which amino acid contains the N-terminal group?
A) histidine
B) proline
C) tyrosine
D) valine
90) In the protein, Asn-Phe-Cys-Lys, which amino acid contains the C-terminal group?
A) asparagine
B) cysteine
C) lysine
D) phenylalanine
91) How many possible tripeptides can be made with the amino acids: tyrosine, histidine, and cysteine,
each used only once?
A) 3
B) 4
C) 6
D) 9

92) Which two amino acids form the dipeptide below?
A) asparagine and proline
B) glycine and cysteine
C) serine and alanine
D) threonine and valine
93) Which of the following notations represents the dipeptide shown below?
A) Phe-Thr
B) Thr-Phe
C) Ser-Tyr
D) Tyr-Ser

94) Which of the following notations represents the polypeptide shown below?
A) Gly-Ala-Ser
B) Gly-Val-Thr
C) Ser-Ala-Gly
D) Thr-Val-Gly
95) How many peptide bonds are present in the polypeptide shown below?
A) one
B) two
C) three
D) four
96) A partial hydrolysis of a protein yielded the following fragments: Ser-Gly, Gly-Ala, Gly-Ala-Val, Val-
Leu, Ala-Val. What is the sequence of amino acids in the polypeptide?
A) Gly-Leu-Ser-Ala-Val
B) Leu-Ala-Gly-Val-Ser
C) Ser-Gly-Ala-Val-Leu
D) Val-Ala-Gly-Ser-Leu

97) Hemoglobin is a protein molecule which contains 4 iron atoms. If the mass percent of iron is 0.342%,
what is the molar mass of hemoglobin?
A) 1020 g/mol
B) 16,300 g/mol
C) 65,300 g/mol
D) none of these
98) Which of the following statements about benzene is true? Benzene
A) contains 3 long bonds and 3 short bonds.
B) is a puckered, six-membered ring.
C) is unusually stable because of delocalized pi electrons.
D) readily undergoes addition reactions.
99) What type of hybridization does a carbon atom have in a six-membered aromatic ring?
A) sp
B) sp2
C) sp3
D) sp3d
100) Which reaction is not characteristic of aromatic compounds?
A) aromatic ring has 4n pi electrons
B) aromatic ring is planar
C) aromatic ring has 4n + 2 pi electrons
D) aromatic ring has p orbitals that overlap
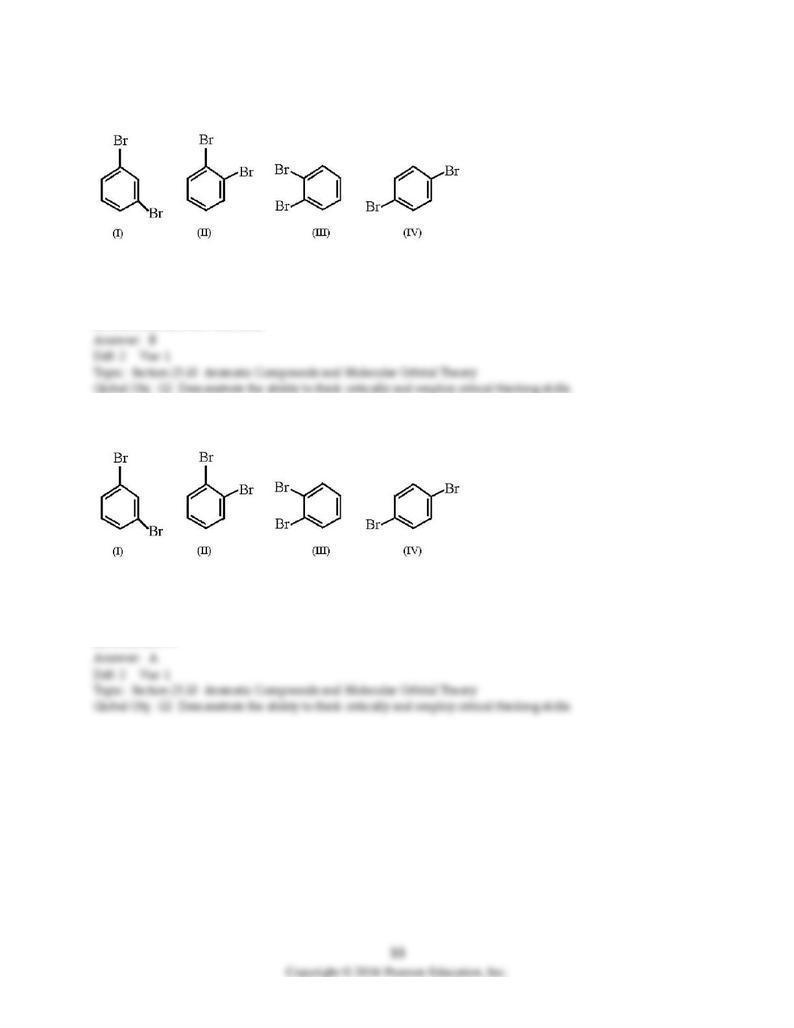
101) Which structures represent the same dibromobenzene compound?
A) I and IV
B) II and III
C) I, II, and III
D) All structures are identical.
102) Which structure is 1,2-dibromobenzene?
A) structure I
B) structure II
C) structure III
D) structure IV

103) Which structure is 1,4-dibromobenzene?
A) structure I
B) structure II
C) structure III
D) structure IV
104) Which one of the following amine bases does not appear in DNA?
A) adenine
B) cytosine
C) guanine
D) uracil
105) The one cyclic amine base that occurs in DNA but not in RNA is
A) cystine.
B) guanine.
C) thymine.
D) uracil.

106) Which of the following is not part of a nucleotide?
A) cyclic amine base
B) fatty acid
C) phosphoric acid
D) a sugar
107) The attractive force between the cylic amine bases in DNA is
A) disulfide bridges.
B) hydrogen bonding.
C) hydrophobic stacking.
D) ionic interactions of salt bridges.
108) What is the sugar component in RNA called?
A) fructose
B) galactose
C) glucose
D) ribose
109) What is the backbone of the nucleic acid constructed of?
A) alternating sugar phosphate groups
B) amine bases
C) glucose polymers
D) polyamides

110) The nucleotide sequence, T-A-G, stands for
A) threonine-alanine-glutamine.
B) thymine-adenine-guanine.
C) tyrosine-asparagine-glutamic acid.
D) None of these.
111) Which of the following bases pairs with adenine in DNA?
A) cytosine
B) guanine
C) thymine
D) uracil
112) What is the complementary DNA sequence to ATATGGTC?
A) CGCGTTGA
B) GCGCAACT
C) TATACCAG
D) TUTUCCAG
113) What is the complementary mRNA sequence to GCTATACCGT?
A) ATCGCGTTAC
B) CGAUAUGGCA
C) TUGCGCUUTG
D) UTCGCGTTUC

114) The base sequences in mRNA that code for particular amino acids are Trp: UGG, Phe: UUU, and
Glu: GAA. What is the peptide that is produced from the DNA sequence: CTTACCAAA?
A) Glu-Phe-Trp
B) Glu-Trp-Phe
C) Phe-Trp-Glu
D) Trp-Glu-Phe
115) DNA has a shape of a
A) tetrahedral molecule.
B) single helix.
C) double helix.
D) linear molecule.
116) What is the H-C-H bond angle?
A) 90°
B) 109.5°
C) 120°
D) 180°
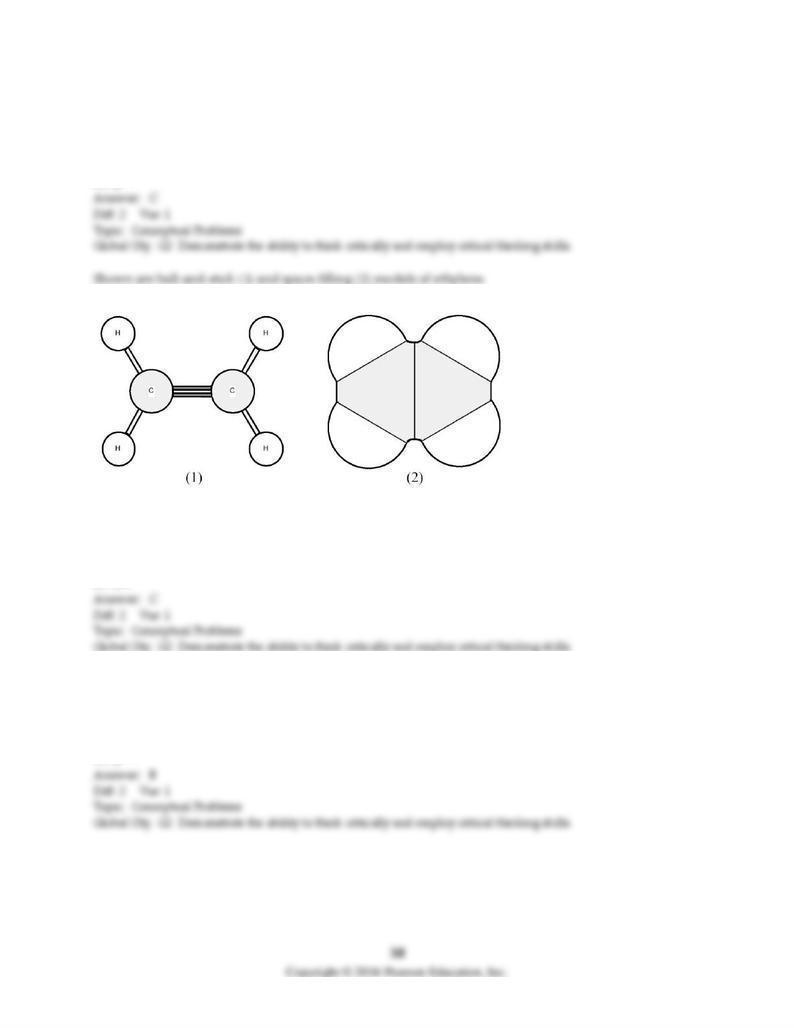
117) What hybrid orbitals are used by carbon to form covalent bonds with hydrogen?
A) sp
B) sp2
C) sp3
D) sp3d
118) What is the H-C-H bond angle?
A) 90°
B) 109.5°
C) 120°
D) 180°
119) What hybrid orbitals are used by carbon to form covalent bonds with hydrogen?
A) sp
B) sp2
C) sp3
D) sp3d
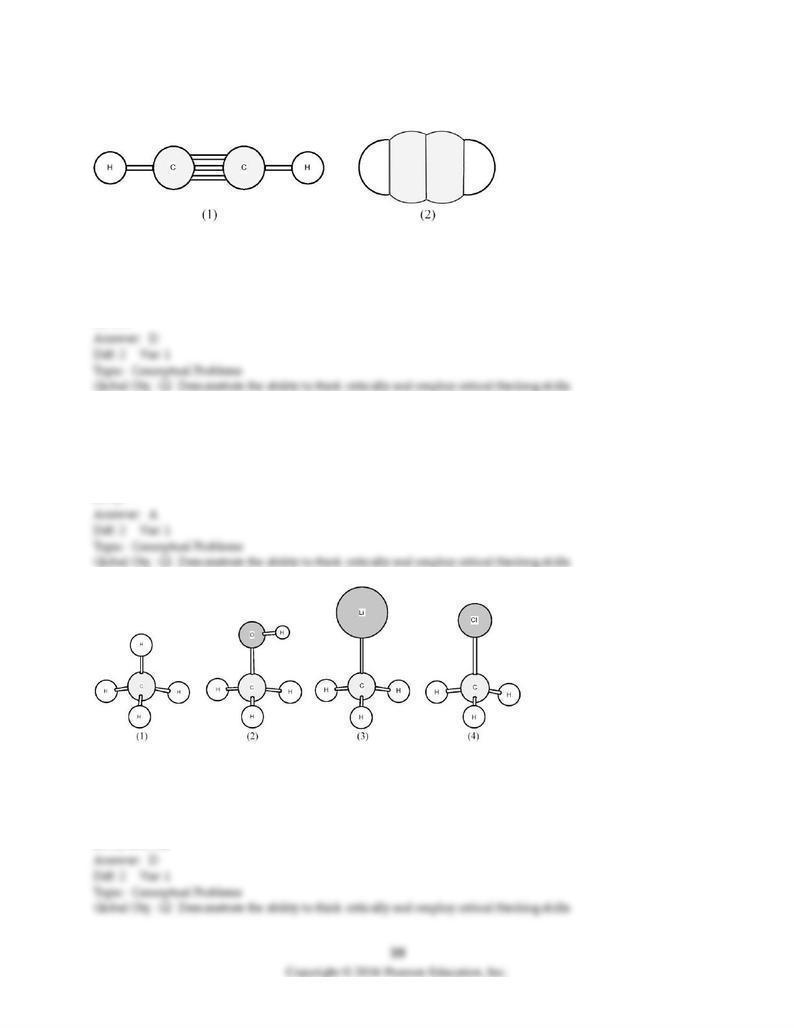
Shown are ball-and-stick (1) and space-filling (2) models of acetylene.
120) What is the H-C-C bond angle?
A) 90°
B) 109.5°
C) 120°
D) 180°
121) What hybrid orbitals are used by carbon to form covalent bonds with hydrogen?
A) sp
B) sp2
C) sp3
D) sp3d
122) Which of these molecules above have a partial positive charge (δ+) on the carbon atom?
A) only (1)
B) only (3)
C) (1) and (3)
D) (2) and (4)
123) Which of these molecules above have a large partial negative charge (δ-) on the carbon atom?
A) only (1)
B) only (3)
C) (1) and (3)
D) (2) and (4)
124) In the figure above, which structures are identical?
A) All are identical.
B) None are identical.
C) only (1) and (4)
D) only (1), (3), and (4)

125) In the figure above, which are isomers of compound (1)?
A) All are isomers of compound (1).
B) None are isomers of compound (1).
C) only (2) and (3)
D) only (4)
126) In the figure above, which are branched hydrocarbons?
A) All are branched hydrocarbons.
B) None are branched hydrocarbons.
C) only (1) and (4)
D) only (1), (2), and (4)
127) Which of the compounds shown above is correctly identified by the name pentane according to the
IUPAC system of naming compounds?
A) all
B) none
C) only (2)
D) only (3)
128) Which of the compounds shown above is correctly identified by the name 2-methylbutane?
A) all are 2-methylbutane
B) none
C) only (1)
D) only (1) and (4)

129) Which of the compounds shown above is correctly identified by the name 3-methylbutane?
A) all
B) none
C) only (4)
D) only (1) and (4)
130) Which of the compounds shown above is correctly identified by the name 2, 2-dimethylpropane?
A) all
B) none
C) only (2)
D) only (1), (2), and (4)
131) Which of the compounds shown above is correctly identified by the name 3, 3-dimethylpropane?
A) all
B) none
C) only (4)
D) only (1) and (4)
132) What is the IUPAC name of the following alkane?
A) decane
B) 2, 4-dimethyloctane
C) 5, 7-dimethyloctane
D) 1, 1, 3-trimethylheptane
133) What is the IUPAC name of the following alkane?
A) 2,5-dimethylhexane
B) octane
C) 1,1,4,4-tetramethylbutane
D) 1,1,4-trimethylpentane
134) Which are isomers?
A) All are isomers.
B) (1) and (2); (3) and (4)
C) (1) and (3); (2) and (4)
D) (1) and (4); (2) and (3)
135) To which family does this organic compound belong?
A) alkane
B) alkene
C) alkyne
D) arene
136) To which family does this organic compound belong?
A) alkane
B) alkene
C) alkyne
D) arene
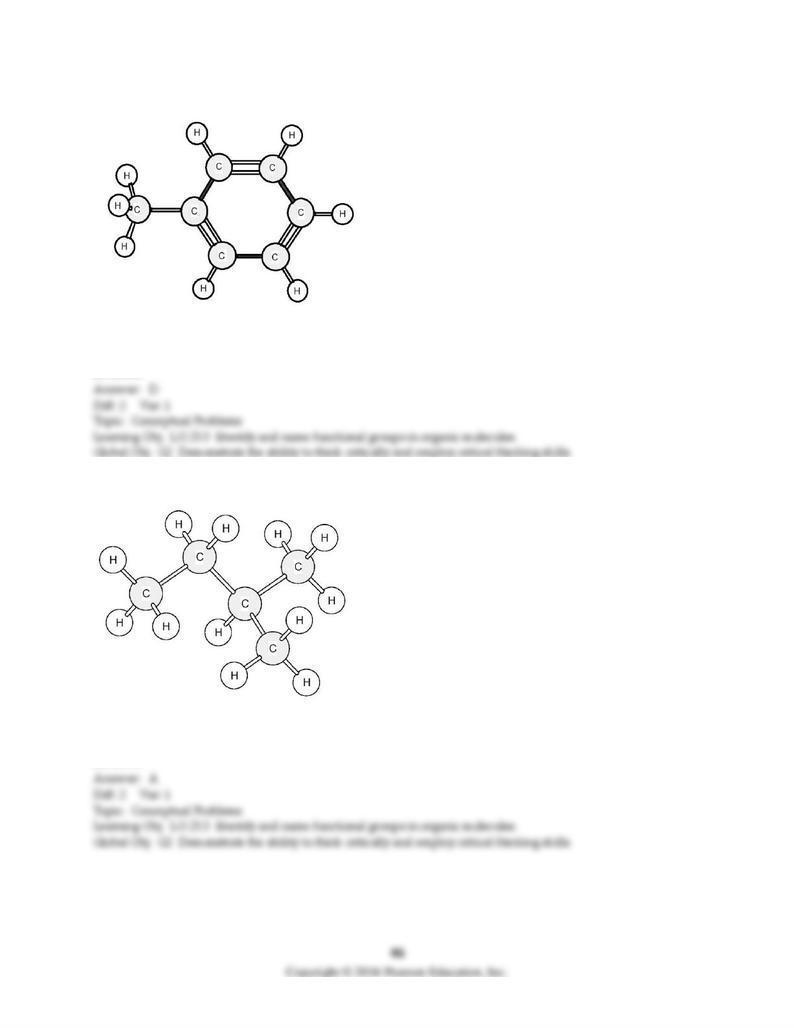
137) To which family does this organic compound belong?
A) alkane
B) alkene
C) alkyne
D) arene
138) To which family does this organic compound belong?
A) alkane
B) alkene
C) alkyne
D) arene
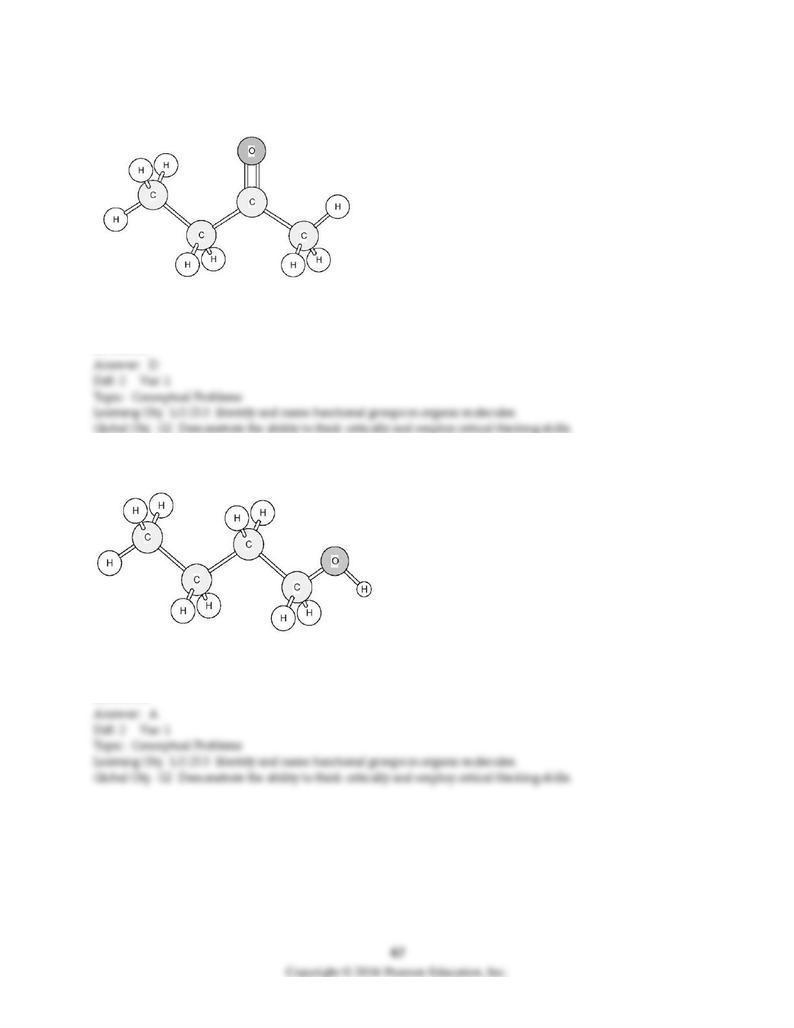
139) To which family does this organic compound belong?
A) alcohol
B) aldehyde
C) ether
D) ketone
140) To which family does this organic compound belong?
A) alcohol
B) aldehyde
C) ether
D) ketone
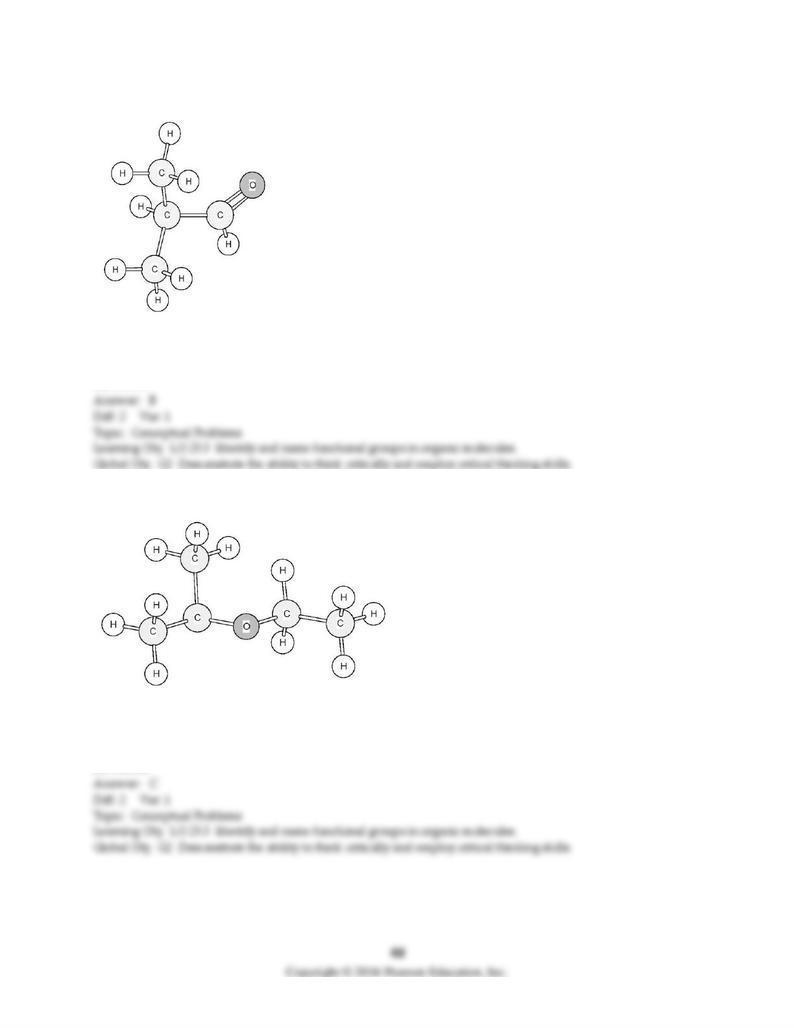
141) To which family does this organic compound belong?
A) alcohol
B) aldehyde
C) ether
D) ketone
142) To which family does this organic compound belong?
A) alcohol
B) aldehyde
C) ether
D) ketone
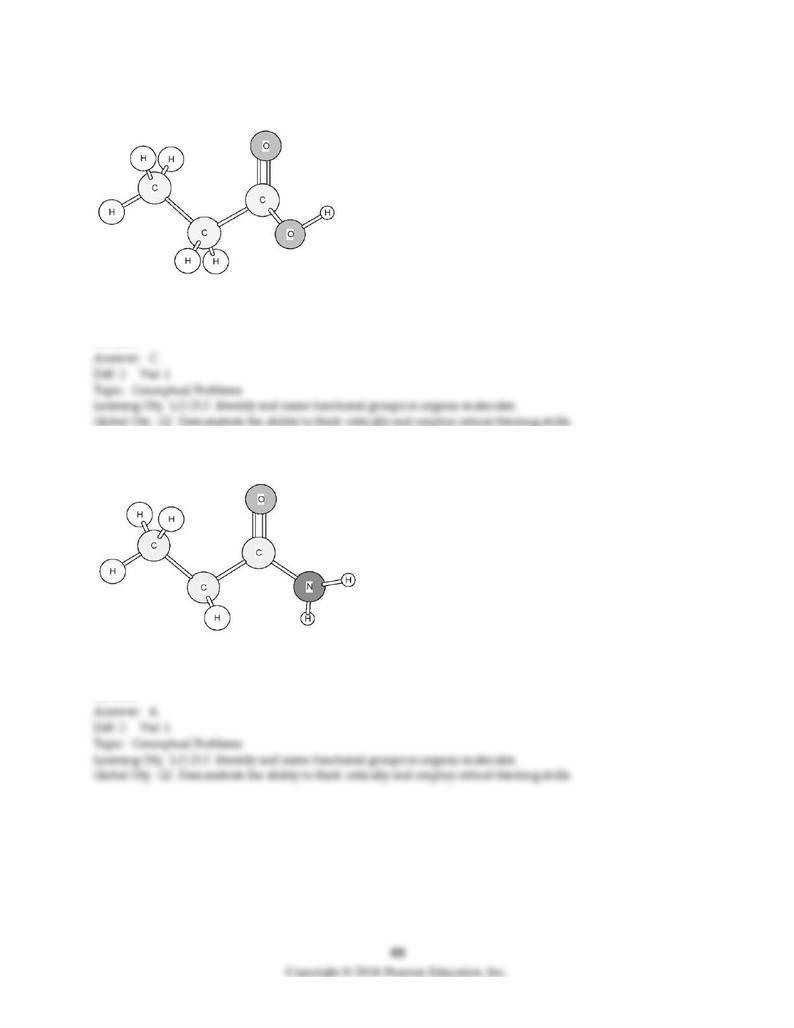
143) To which family does this organic compound belong?
A) amide
B) amine
C) carboxylic acid
D) ester
144) To which family does this organic compound belong?
A) amide
B) amine
C) carboxylic acid
D) ester

145) To which family does this organic compound belong?
A) amide
B) amine
C) carboxylic acid
D) ester
146) To which family does this organic compound belong?
A) amide
B) amine
C) carboxylic acid
D) ester
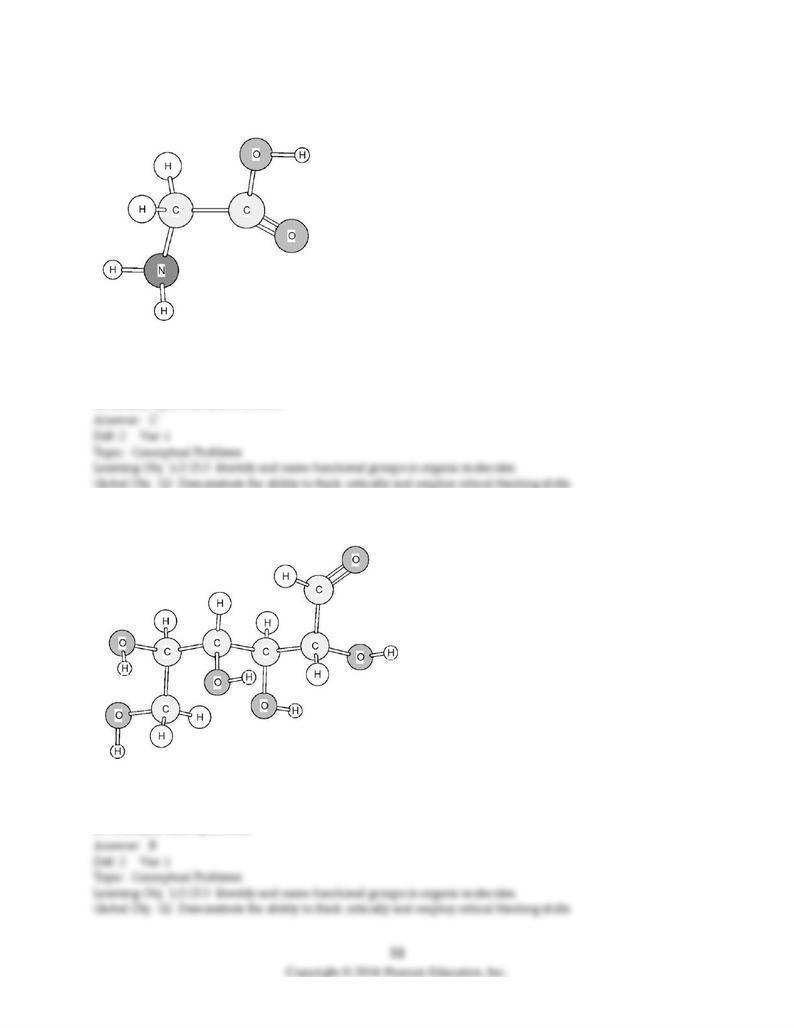
147) Identify the functional groups in the following compound.
A) alcohol and ketone
B) alcohol, amine, and ketone
C) amine and carboxylic acid
D) amine, hydroxide, and ketone
148) Identify the functional groups in the following compound.
A) alcohol
B) alcohol and aldehyde
C) alcohol and ketone
D) aldehyde and hydroxide
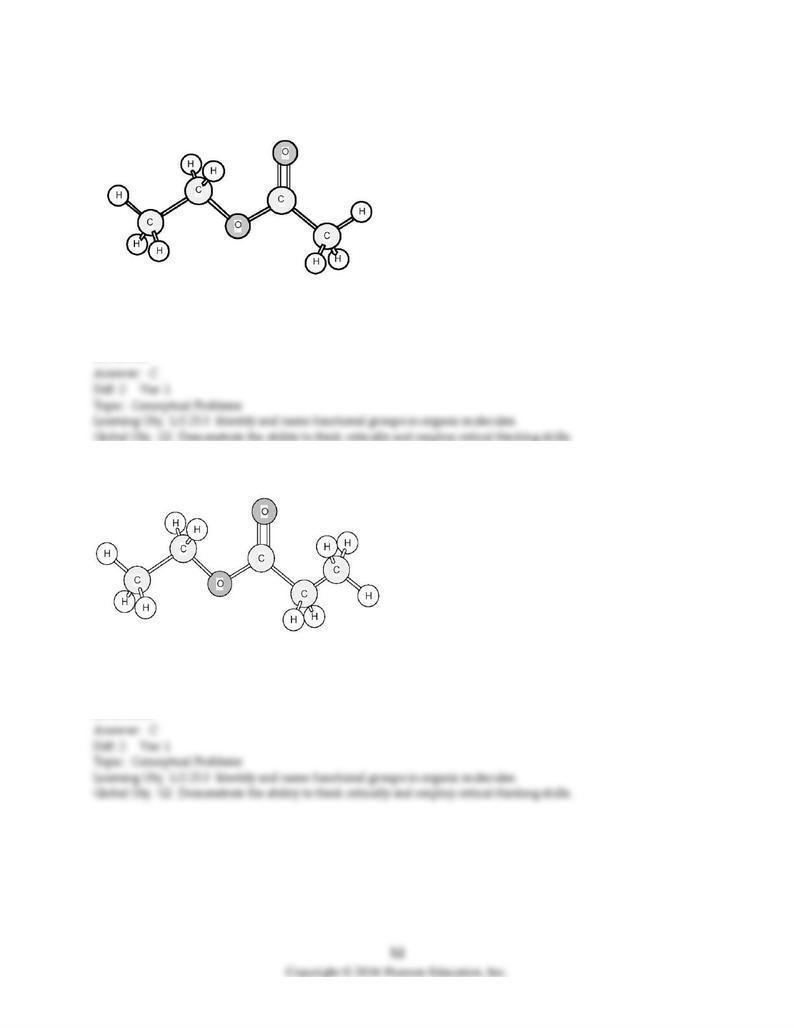
149) Identify the functional group.
A) carboxylic acid
B) aldehyde
C) ester
D) ketone
150) Identify thefunctional group.
A) ether
B) aldehyde
C) ester
D) alcohol
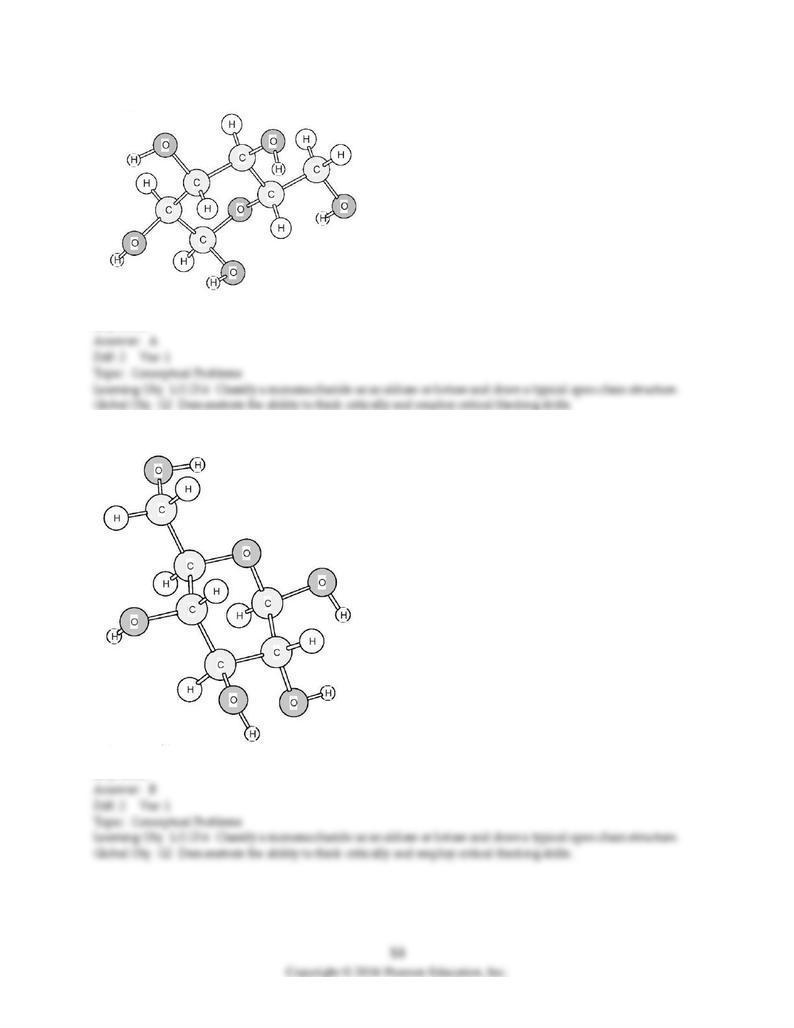
151) Is the following model of glucose in the α form or the β form?
A) α form
B) β form
152) Is the following model of glucose in the α form or the β form?
A) α form
B) β form

153) Identify the amino acid shown below.
A) Alanine (Ala)
B) Glycine (Gly)
C) Isoleucine (Ile)
D) Leucine (Leu)

154) Identify the amino acid shown below.
A) Alanine (Ala)
B) Glycine (Gly)
C) Isoleucine (Ile)
D) Leucine (Leu)

155) Identify the amino acid shown below.
A) Alanine (Ala)
B) Glycine (Gly)
C) Isoleucine (Ile)
D) Leucine (Leu)

156) Identify the amino acid shown below.
A) Alanine (Ala)
B) Glycine (Gly)
C) Isoleucine (Ile)
D) Leucine (Leu)

157) Identify the amino acid shown below.
A) Methionine (Met)
B) Phenylalanine (Phe)
C) Proline (Pro)
D) Tryptophan (Trp)

158) Identify the amino acid shown below.
A) Methionine (Met)
B) Phenylalanine (Phe)
C) Proline (Pro)
D) Tryptophan (Trp)

159) Identify the amino acid shown below.
A) Methionine (Met)
B) Phenylalanine (Phe)
C) Proline (Pro)
D) Tryptophan (Trp)

160) Identify the amino acid shown below.
A) Methionine (Met)
B) Phenylalanine (Phe)
C) Proline (Pro)
D) Tryptophan (Trp)
161) Identify the amino acid shown below.
A) Cysteine (Cys)
B) Serine (Ser)
C) Threonine (Thr)
D) Tyrosine (Tyr)

162) Identify the amino acid shown below.
A) Cysteine (Cys)
B) Serine (Ser)
C) Threonine (Thr)
D) Tyrosine (Tyr)

163) Identify the amino acid shown below.
A) Cysteine (Cys)
B) Serine (Ser)
C) Threonine (Thr)
D) Tyrosine (Tyr)

164) Identify the amino acid shown below.
A) Cysteine (Cys)
B) Serine (Ser)
C) Threonine (Thr)
D) Tyrosine (Tyr)

165) Identify the amino acid shown below.
A) Asparagine (Asn)
B) Aspartic acid (Asp)
C) Glutamic acid (Glu)
D) Glutamine (Gln)
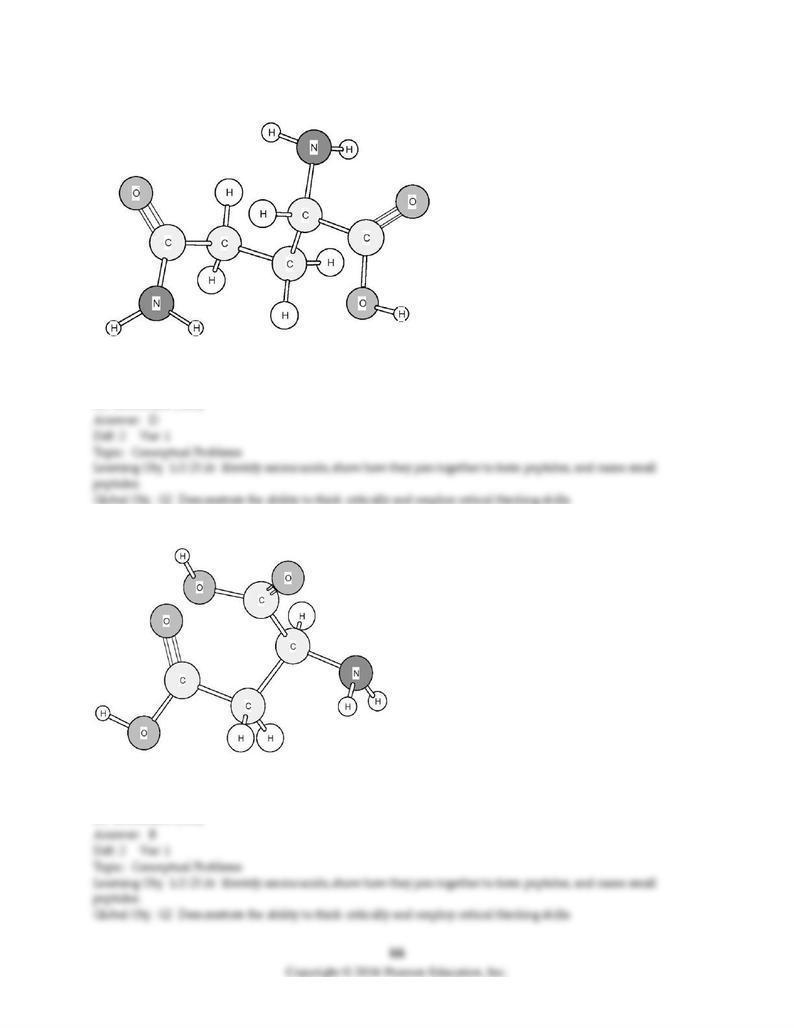
166) Identify the amino acid shown below.
A) Asparagine (Asn)
B) Aspartic acid (Asp)
C) Glutamic acid (Glu)
D) Glutamine (Gln)
167) Identify the amino acid shown below.
A) Asparagine (Asn)
B) Aspartic acid (Asp)
C) Glutamic acid (Glu)
D) Glutamine (Gln)
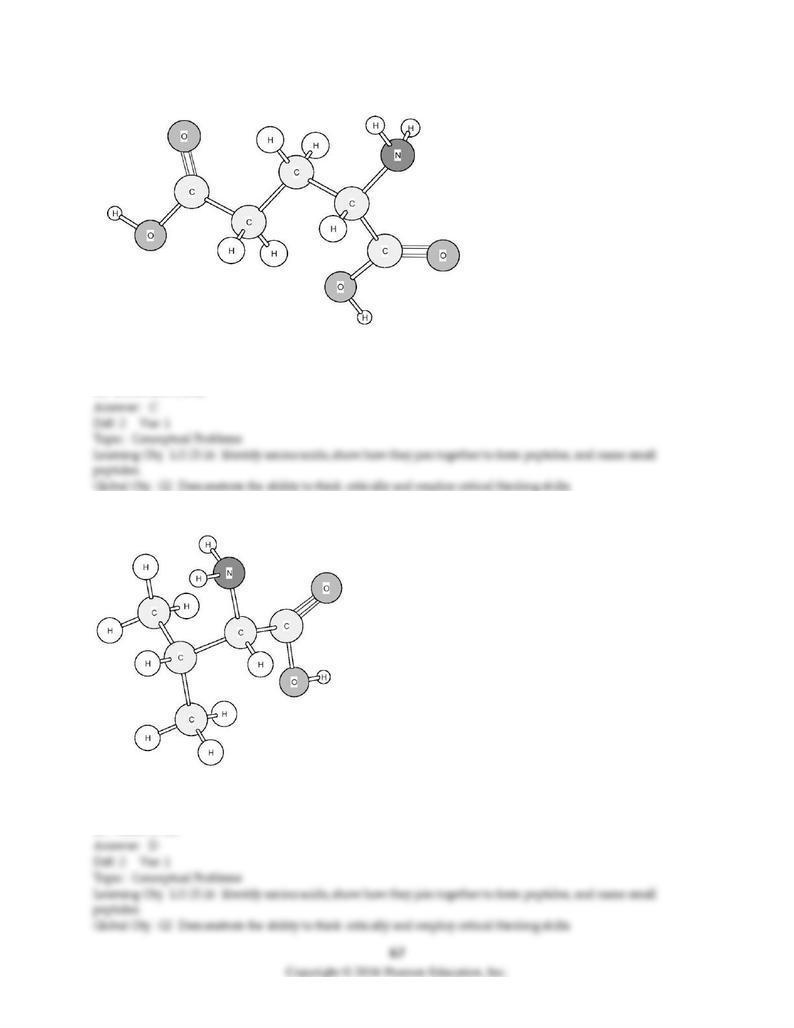
168) Identify the amino acid shown below.
A) Asparagine (Asn)
B) Aspartic acid (Asp)
C) Glutamic acid (Glu)
D) Glutamine (Gln)
169) Identify the amino acid shown below.
A) Arganine (Arg)
B) Histidine (His)
C) Lysine (Lys)
D) Valine (Val)
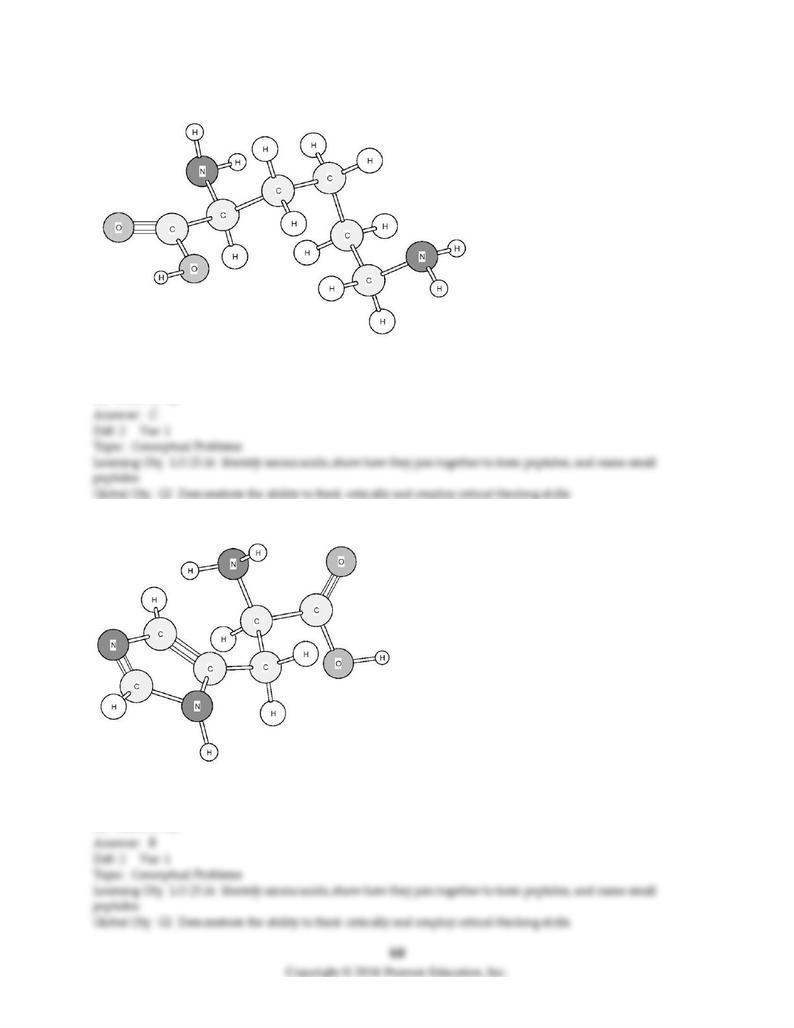
170) Identify the amino acid shown below.
A) Arganine (Arg)
B) Histidine (His)
C) Lysine (Lys)
D) Valine (Val)
171) Identify the amino acid shown below.
A) Arganine (Arg)
B) Histidine (His)
C) Lysine (Lys)
D) Valine (Val)
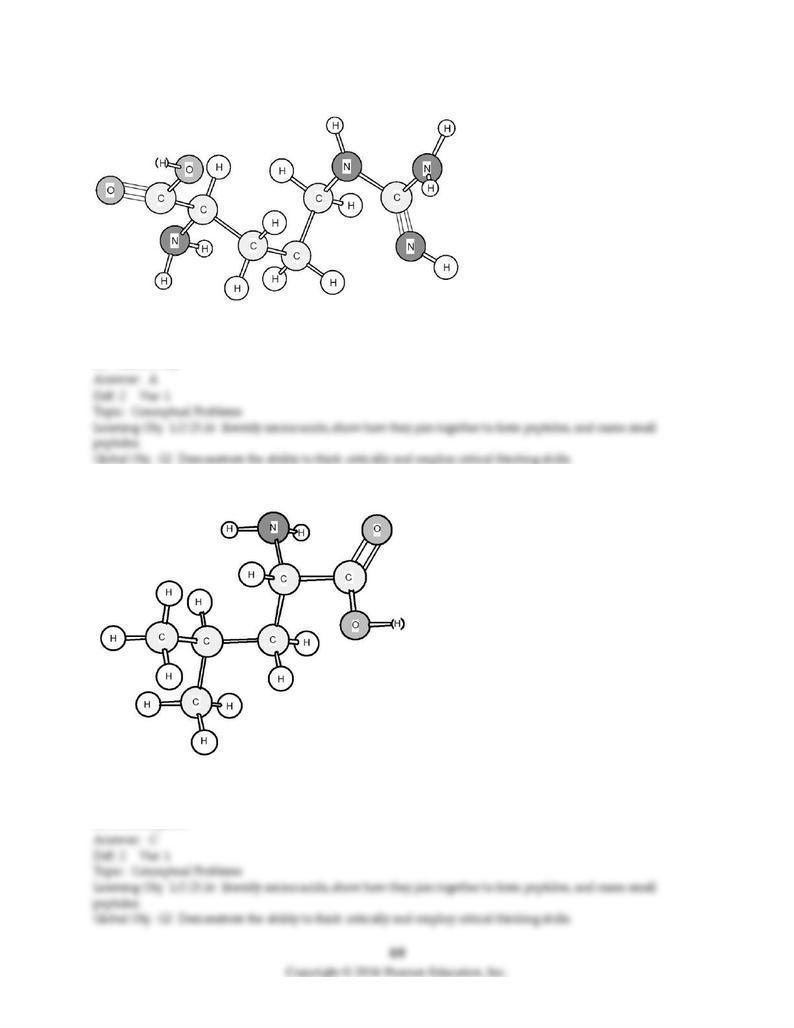
172) Identify the amino acid shown below.
A) Arganine (Arg)
B) Histidine (His)
C) Lysine (Lys)
D) Valine (Val)
173) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
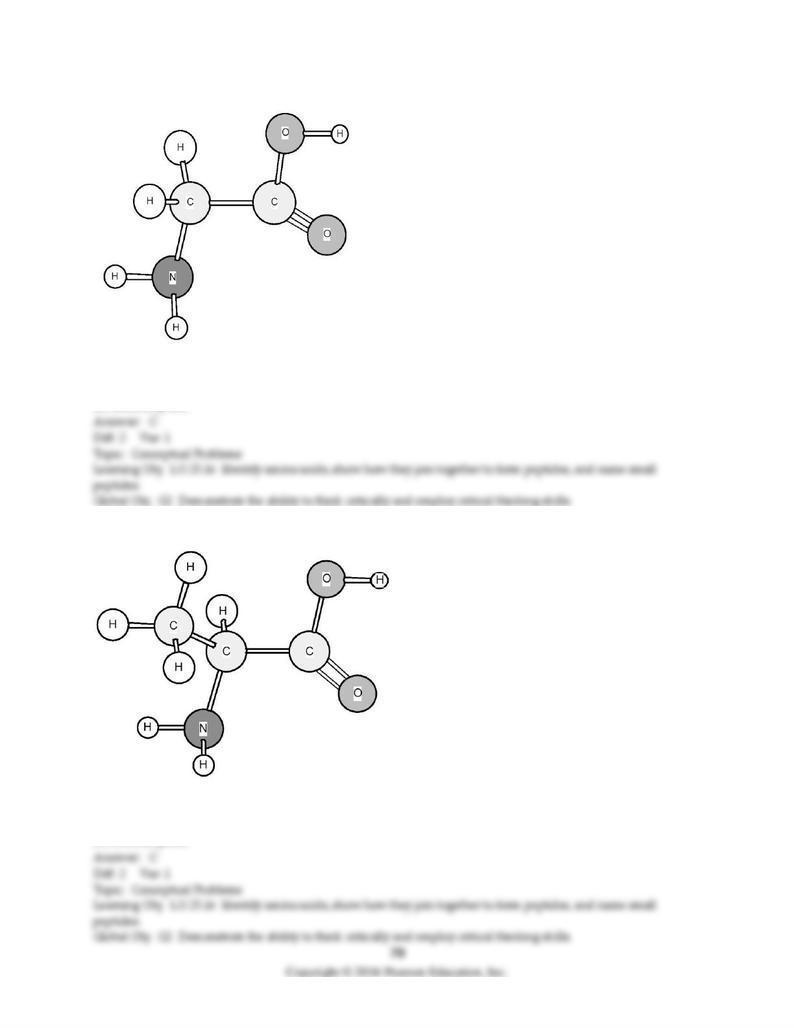
174) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
175) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

176) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

177) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

178) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
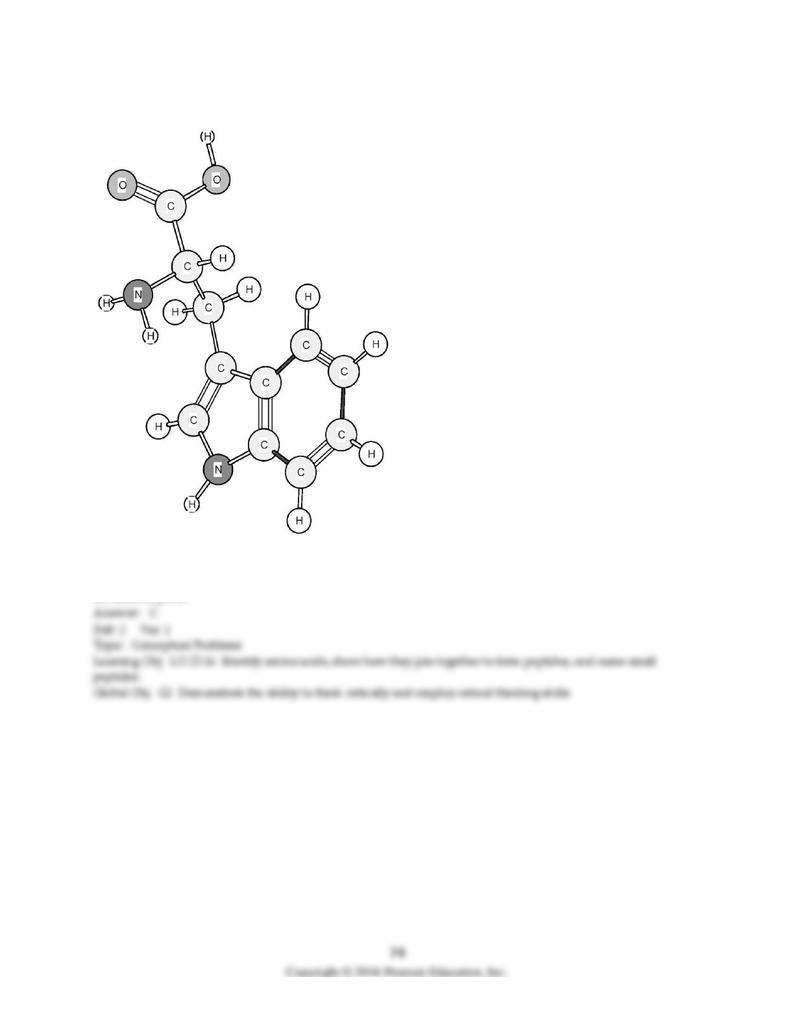
179) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

180) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

181) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

182) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

183) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
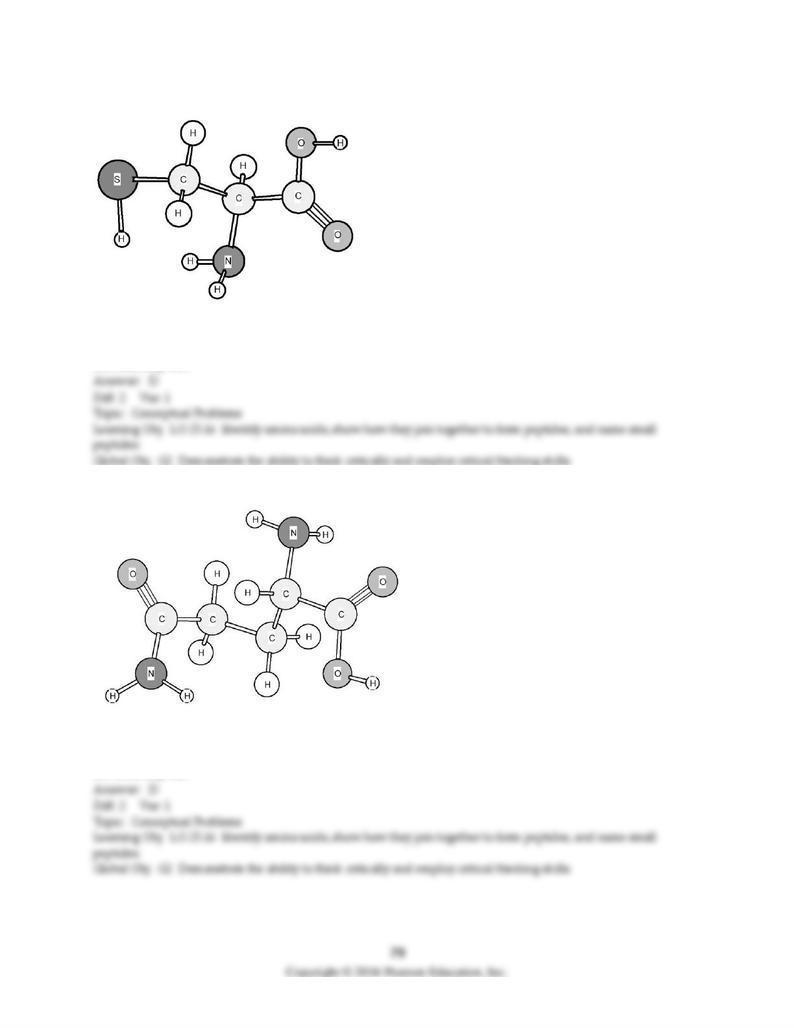
184) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
185) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
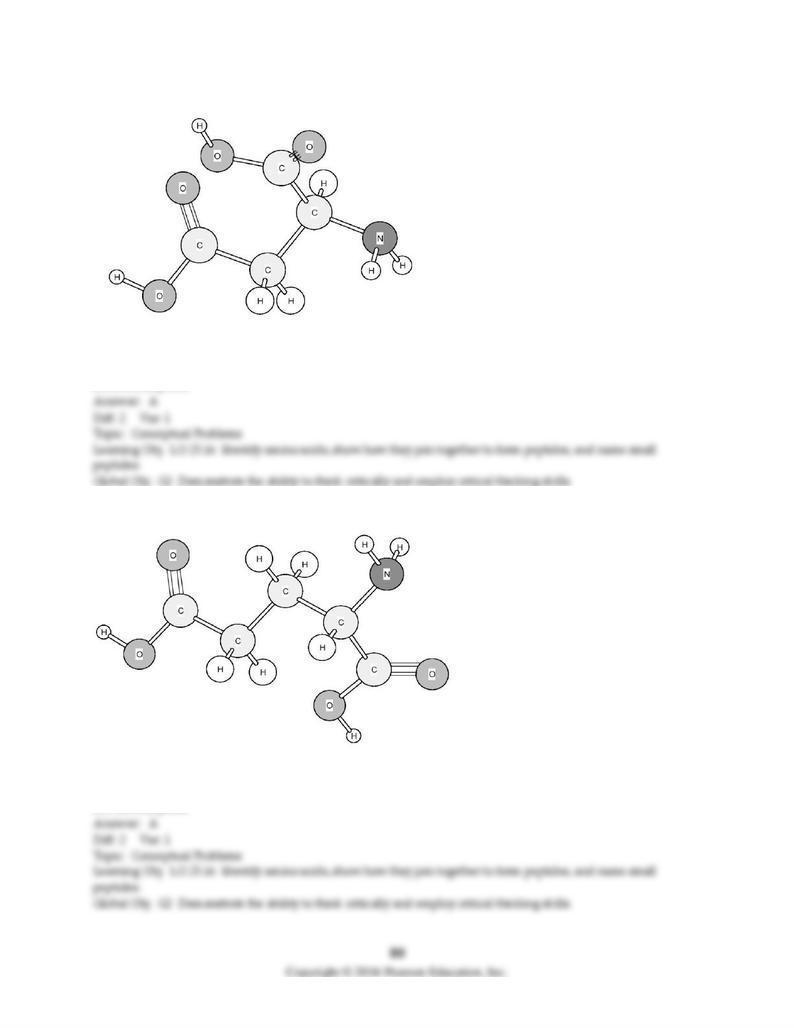
186) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
187) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

188) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
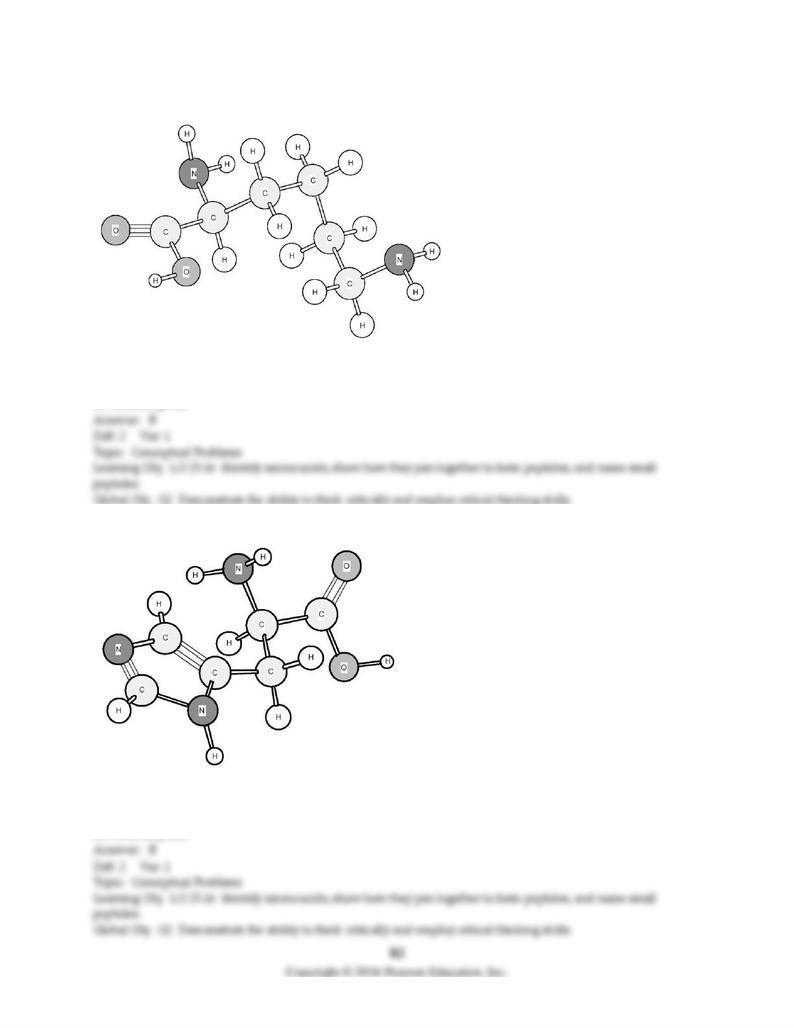
189) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.
190) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

191) Based on the structure of its side chains, the amino acid shown below can be classified as
A) acidic.
B) basic.
C) neutral, non-polar.
D) neutral, polar.

192) Identify the following dipeptide.
A) Ser-Ala
B) Ser-Leu
C) Ser-Phe
D) Ser-Val

193) Identify the following dipeptide.
A) Ser-Ala
B) Ser-Leu
C) Ser-Phe
D) Ser-Val

194) Identify the following amine base found in nucleic acids.
A) Adenine (A)
B) Cytosine (C)
C) Thymine (T)
D) Uracil (U)

195) Identify the following amine base found in nucleic acids.
A) Adenine (A)
B) Cytosine (C)
C) Thymine (T)
D) Uracil (U)
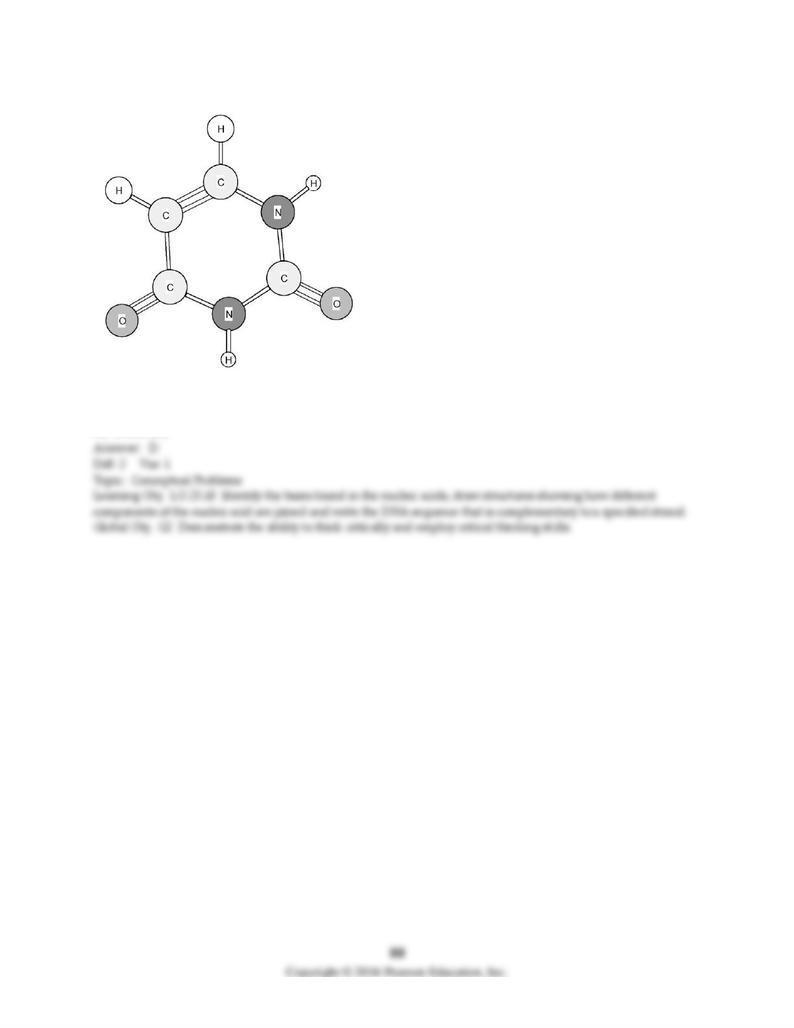
196) Identify the following amine base found in nucleic acids.
A) Adenine (A)
B) Cytosine (C)
C) Thymine (T)
D) Uracil (U)

197) Identify the following amine base found in nucleic acids.
A) Adenine (A)
B) Cytosine (C)
C) Thymine (T)
D) Uracil (U)

23.2 Algorithmic Questions
1) How many isomers are there for C4H10?
A) 2
B) 3
C) 4
D) 5
2) When butane, , reacts with oxygen to produce carbon dioxide and water, 2880 kJ of heat are
released per mole of methane. How many kJ of heat are released per gram of oxygen used?
A) 6.92 kJ/g
B) 13.8 kJ/g
C) 585 kJ/g
D) 1170 kJ/g
3) Which one of the following molecules is the most polar?
A) acetylene
B) benzoic acid
C) diethylether
D) 2, 3-dimethylhexane
4) What is an example of the proper number of bonds to carbon?
A) two double bonds
B) one double bond + one triple bond
C) three single bonds
D) five single bonds

5) Benzene has a formula of
A) C3H8.
B) C6H10.
C) C6H6.
D) C8H16.
6) Which one of the following compounds is an alcohol?
A) CH3OCH3
B) CH3COCH2CH3
C) (CH3)2CHOH
D) CH3CO2CH3
7) The compound that does not contain oxygen is
A) aldehyde.
B) ester.
C) amide.
D) aromatic.
8) Identify the compound that contains two oxygens.
A) aldehyde
B) alkyne
C) amine
D) carboxylic acid

9) Which compound is a ketone?
A) butanal
B) propanoic acid
C) 2-butanone
D) butane
10) Which of the following molecules is the most highly oxidized, in other words, the one with the most
number of C-O bonds?
A) acetaldehyde
B) ethanoic acid
C) dimethyl ether
D) ethylene
11) Which one of the following contains a polar carbonyl group, C=O?
A) a carboxylic acid
B) an alkyne
C) an amine
D) an alkane
12) Which of the following pairs have the most similar chemical properties?
A) alkynes and esters
B) alkenes and aromatics
C) amines and carboxylic acids
D) ketones and aldehydes

13) Identify the compound that is an ester.
A) propyl propanoate
B) 3-hexanone
C) ethanoic acid
D) dimethyl ether
14) Identify the compound that is an aldehyde.
A) ethyl pentanoate
B) butanal
C) dimethyl ether
D) 3-pentanone
15) Identify the amine.
A) CH3CH2COOCH3
B) CH3CH2CH=CHCH3
C) CH3CH2OH
D) CH3CH2H2
16) Identify the ether.
A) CH3CH2OCH3
B) CH3CH2CH=CH2
C) CH3CH2OH
D) CH3CH2NHCH3

17) Which compound contains a nitrogen and a C=O?
A) ether
B) amide
C) carboxylic acid
D) alkene
18) Which compound contains only carbons and hydrogens?
A) methyl ethanoate
B) acetylene
C) 2-butanone
D) ethyl amine
19) Identify the compound that contains a carbon-carbon double bond.
A) 2-pentyne
B) 1-pentene
C) methyl propanoate
D) methanol
20) Which compound is a saturated hydrocarbon?
A) benzene
B) cyclohexene
C) 2-methylpentane
D) propylene

21) Which compound is a saturated hydrocarbon?
A) benzene
B) cycloheptene
C) 2-methylhexane
D) butyne
22) What is the IUPAC name for the continuous carbon chain of C4H10?
A) butane
B) methane
C) butene
D) octyne
23) Which name is not correct?
A) 2, 2-dimethylbutane
B) 2, 3-dimethyloctane
C) 2, 6, 6-trimethylheptane
D) 2, 3, 3-trimethyloctane
24) None of the listed compounds contains a ring. Which could have one carbon-carbon double bond?
A) C9H20
B) C2H2
C) C3H6
D) All of these

25) None of the listed compounds contains a ring. Which could have one carbon-carbon triple bond?
A) C3H8
B) C2H2
C) C10H20
D) All of these
26) None of the listed compounds contains a ring. Which has at least one singly-bonded carbon atom?
A) C5H12
B) C4H6
C) C6H12
D) All of these
27) Which one of the following molecules can rotate freely around its carbon-carbon bond?
A) benzene
B) cyclopropane
C) pentane
D) ethene
28) Which of the following is a correct name for an alkyne?
A) 3-methyl-2-octyne
B) 8-methyl-3-octyne
C) 2, 2-dimethyl-4-pentyne
D) 3, 3-dimethyl-1-heptyne

29) Name the product obtained from the addition of chlorine to 3-methyl-1-octene.
A) 1, 1-dichlorooctane
B) 1, 2-dichloro-3-methylbutane
C) 1, 2-dichlorononane
D) trans-1, 2-dichlorooctene
30) The smallest alkyne is called
A) ethene.
B) propene.
C) 1-butyne.
D) acetylene.
31) Identify the saturated compound, assuming none of the structures are rings.
A) C3H6
B) C3H8
C) alkyne
D) C7H12
32) Which name is not correct?
A) 1, 1-dimethylcyclopentane
B) 1, 2-dimethylcyclopentane
C) 1, 3-dimethylcyclooctane
D) 1, 4-dimethylcyclopentane

33) Which of the following does not contain a carbonyl group, C=O?
A) an alkene
B) a ketone
C) an amide
D) an ester
34) Which ending on a ketone or aldehyde indicates a sugar?
A) -ene
B) -one
C) ether
D) -ose
35) Which of the following is not a geometric (cis-trans) isomer?
A) 1, 1-dichloro-1-pentene
B) 1, 2-dichloro-1-hexene
C) 2-hexene
D) 2-chloro-2-heptene
36) Identify the alkene that cannot be drawn cis and trans.
A) 2-pentene
B) 3-hexene
C) 4-octene
D) 1-hexene

37) Identify the compounds that have at least one sp2 hybridized carbon.
A) 1-pentyne
B) propane
C) 2-butene
D) 3-chloropentane
38) Which of the following atoms is expected to be sp2 hybridized?
A) C in C2H6
B) C-H in C3H4
C) O in CH3CH2OH
D) O in CHO
39) Which one of the following amino acids does not contain a basic side chain?
A) arginine
B) histidine
C) lysine
D) glutamine
40) Which one of the following amino acids contains a polar side chain?
A) valine
B) glycine
C) cysteine
D) alanine

41) Which one of the following amino acids contains a hydrophobic side chain?
A) alanine
B) asparagine
C) cysteine
D) glutamine
42) In the protein, Val-Tyr-His-Pro, which amino acid contains the N-terminal group?
A) histidine
B) proline
C) tyrosine
D) valine
43) In the protein, Asn-Phe-Cys-Lys, which amino acid contains the C-terminal group?
A) aspargine
B) cysteine
C) lysine
D) phenylalanine
44) All reactions in the human body are catalyzed by
A) carboxylic acids.
B) alcohols.
C) proteins.
D) alkanes.

45) Keratin is found in
A) cells.
B) bird feathers.
C) muscles.
D) DNA.
46) The human body can synthesize ________ amino acids.
A) 3
B) 11
C) 8
D) 15
47) The human body is thought to contain ________ different kinds of proteins.
A) 90,000
B) 150,000
C) 200,000
D) 110,000
48) Approximately what mass percent of a body's dry weight consists of proteins?
A) 5%
B) 40%
C) 50%
D) 70%

49) A peptide bond links
A) an alcohol and a ketone.
B) an amine and an aldehyde.
C) an amine and a carboxylic acid.
D) an ester and a carboxylic acid.
50) How many alpha amino acids commonly make up the different proteins found in humans?
A) 6
B) 20
C) 22
D) 12
51) Which one of the following amino acids contains a basic side chain?
A) proline
B) cytosine
C) glutamic acid
D) arginine
52) Which of the following amino acids is acidic?
A) alanine
B) aspartic acid
C) glutamine
D) histidine

53) Which one of the following amino acids contains a polar side chain?
A) alanine
B) glycine
C) serine
D) histidine
54) Which of the following amino acids contains a benzyl group?
A) isoleucine
B) proline
C) asparagine
D) tryrosine
55) Which one of the amino acids contains a double ring structure?
A) alanine
B) serine
C) tryptophan
D) histidine
56) Which amino acid has the simplest structure?
A) valine
B) glycine
C) asparagine
D) arginine

57) Which of the following amino acids contains a sulfur in its side chain?
A) aspartic acid
B) glycine
C) cysteine
D) arginine
58) Which one of the following amino acids contains a hydrophobic side chain?
A) leucine
B) cysteine
C) glutamine
D) lysine
59) All but one of the common amino acids contain a -NH2 amino group. Which one contains an -NH-
amino group as part of a ring structure?
A) phenylalanine
B) proline
C) tyrosine
D) arginine
60) In leu-ala-arg-val-tyr-his-pro-gln, the amino acid with the N-terminal is
A) gln.
B) val.
C) leu.
D) tyr.

61) The structure of DNA was proposed by
A) Einstein.
B) Curie.
C) Watson and Crick.
D) Copernicus.
1) In organic molecules the molecular geometry exhibited by carbon is ________ when carbon is bonded
to four atoms by four single bonds, ________ when carbon is bonded to three atoms by one double bond
and two single bonds, and ________ when carbon is bonded to two atoms by one triple bond and one
single bond.
2) The H-C-H bond angles in C2H6, C2H4, and C2H2 are ________, ________, and ________, respectively.
3) Orbitals used by carbon for bonding in C2H6, C2H4, and C2H2 are ________, ________, and ________,
respectively.
4) The C-Br bond in CH3Br is a ________ (nonpolar, polar) covalent bond with a ________ (δ+, δ-) charge
on the carbon atom and a ________ (δ+, δ-) charge on the bromine atom.
5) The C-Br bond in CH3CH2Li is a ________ (nonpolar, polar) covalent bond with a ________ (δ+, δ-)
charge on the carbon atom and a ________ (δ+, δ-) charge on the lithium atom.

6) Organic molecules containing only carbon and hydrogen and having only single bonds are called
________ hydrocarbons, or ________.
7) How many isomers exist for C5H12? Of these isomers, how many are straight-chain isomers and how
many are branched isomers?
8) Do the condensed structures shown below represent the same or different molecules?
(1) CH3CH2CH2CH2CH(CH3)2
(2) CH3
|
CH3CHCH2CH2CH2CH3
9) Do the two condensed structures (1) and (2) below represent the same or different molecules?
CH3 CH3 CH3
| | |
CH3CHCH2CHCH3 CH3CHCH2CHCH3
|
CH3
(1) (2)
10) The number of different monochloro substitution products (C5H11Cl) that can form from the reaction
of 2-methylbutane with Cl2 is ________.

11) The number of different monochloro substitution products (C5H11Cl) that can form from the reaction
of 2,2-dimethylpropane with Cl2 is ________.
12) The combustion of methane has a standard enthalpy of combustion H° = -802 kJ per mole of methane
burned. How many kilojoules of heat are released if 1.28 grams of oxygen are consumed in the reaction?
13) The condensed formula of the smallest molecule that contains the ether functional group is ________.
14) In the dark the reaction of 2-pentene with bromine will form how many different products?
15) C2H6O is the empirical formula for both ethyl alcohol, CH3CH2OH and dimethyl ether, CH3OCH3.
In order to boil CH3CH2OH the strongest intermolecular forces that must be overcome are (London
dispersion, dipole-dipole, hydrogen bonding) forces. In order to boil CH3OCH3 the strongest
intermolecular forces that must be overcome are (London dispersion, dipole-dipole, hydrogen bonding)
forces. Which compound is expected to have the higher boiling point?
16) The molecule H2NCH2CH2CH2OH contains an ________ functional group and an ________
functional group.

17) The molecule CH3CH2CO2CH2CO2H contains an ________ functional group and a ________
functional group.
18) The condensed structures for the two isomers of C4H10 are (1) ________ and (2) ________.
19) The IUPAC name for the alkane shown below is ________.
CH3
|
CH3CHCH2CH2CH2CH3
20) The alkane, 3-ethyl-3-methylhexane contains ________ carbon atoms and ________ hydrogen atoms.
21) (CH3)3C— represents a ________ (normal, secondary, tertiary) butyl group.

22) What is the empirical and molecular formulas of 1-tert-butyl-2-methylcyclodecane?
23) The molecules CH3CH2CH2CH2C CH and CH3CH2CH2CH2CH=CH2, respectively, belong to the
________ family and the ________ family of organic molecules.
24) The cycloalkane represented by a heptagon is named ________ and has the molecular formula
________.
25) Monosaccharides are classified as either ________ or ________.
26) Linoleic acid, a fatty acid, contains ________ carbons and ________ double bonds.
27) Naturally occurring organic molecules that dissolve in nonpolar solvents are called ________ of which
the most plentiful in nature are ________ and ________.

28) The amide bonds that form when two or more amino acids link together are called ________ bonds.
29) Does tyrosine contain an aromatic ring?
30) Cytosine is a base that occurs in ________ (DNA, RNA, DNA and RNA).
31) The four cyclic amine bases that occur in DNA are ________, ________, ________, and ________.
32) The sugar in RNA is ________.