
Chemistry, 7e (McMurry/Fay)
Chapter 8 Covalent Compounds: Bonding Theories and Molecular Structure
8.1 Multiple-Choice Questions
1) Based on VSEPR theory, which should have the smallest XAX bond angle?
A)
B)
C)
D)
2) Which of the following should be nonplanar?
A) only I
B) only II
C) only III
D) I and III

3) What is the molecular geometry of XeF4?
A) seesaw
B) square planar
C) square pyramidal
D) tetrahedral
4) Which of the following should be nonlinear?
A) only I
B) only II
C) only III
D) II and III
5) What is the molecular geometry of CH3-?
A) T-shaped
B) tetrahedral
C) trigonal planar
D) trigonal pyramidal

6) What is the F—B—F bond angle in BF3?
A) less than 109.5°
B) 109.5°
C) 120°
D) greater than 120°
7) What is the molecular geometry of TeF5-?
A) octahedral
B) seesaw
C) square pyramidal
D) trigonal bipyramidal
8) Which of the following best describes CO2? It has a molecular geometry that is
A) linear molecular shape with no lone pairs on the I atom.
B) linear molecular shape with lone pairs on the I atom.
C) non-linear molecular shape with no lone pairs on the I atom.
D) non-linear molecular shape with lone pairs on the I atom.
9) What is the molecular geometry of SCl4?
A) seesaw
B) square planar
C) square pyramidal
D) tetrahedral

10) What is the smallest bond angle in SF6?
A) 60°
B) 90°
C) 109.5°
D) 120°
11) Which of the following is not a valence bond concept?
A) The greater the overlap between the orbitals on two atoms, the stronger the bond.
B) Lone pair electrons are in atomic orbitals or in hybrid atomic orbitals.
C) Atomic orbitals on two atoms may overlap to form antibonding orbitals.
D) A pair of electrons in a bond is shared by both atoms.
12) Which orbital hybridization is associated with a tetrahedral cloud arrangement?
A) sp
B) sp2
C) sp3
D) sp4
13) What is the angle between adjacent sp3 hybrid orbitals?
A) 90°
B) 109.5°
C) 120°
D) 180°

14) Which of the following is not true?
A) The sp3 hybrid orbitals are degenerate.
B) An sp3 hybrid orbital may hold a lone pair of electrons.
C) An sp3 hybrid orbital may form a sigma bond by overlap with an orbital on another atom.
D) An sp3 hybrid orbital may form a pi bond by overlap with an orbital on another atom.
15) The number of sp2 hybrid orbitals on the carbon atom in CO32- is
A) one.
B) two.
C) three.
D) four.
16) A triple bond is generally composed of
A) three π bonds.
B) two π bonds and one σ bond.
C) one π bond and two σ bonds.
D) three σ bonds.
17) The orbital hybridization on the central carbon atom in CH3CCH is
A) sp.
B) sp2.
C) sp3.
D) sp4.

18) Which of the following would be expected to have sp2 hybridization on atom A?
A) II
B) I and III
C) I, II, and III
D) I and IV
19) The C O bond in COCl2 can be described as
A) a σ bond and a π bond, both involving sp hybrid orbitals on C.
B) a σ bond involving an sp hybrid orbital on C and a π bond involving a p orbital on C.
C) a σ bond and a π bond, both involving sp2 hybrid orbitals on C.
D) a σ bond involving an sp2 hybrid orbital on C and a π bond involving a p orbital on C.
20) Which type of bond produces a charge cloud with a nodal plane containing the bond axis?
A) σ
B) π
C) σ and π
D) neither σ nor π

21) Which atomic orbitals are involved in bonding and which as lone pair orbitals for N2H2?
H — = — H
A) bonding: s on H, sp2 on N lone pair: p on N
B) bonding: sp2 on both H and N lone pair: p on N
C) bonding: s on H, p on N lone pair: sp2 on N
D) bonding: s on H, sp2 and p on N lone pair: sp2 on N
22) What is the hybridization on the N atom in NO2- and in NO3-?
A) sp2 for NO2- and sp3 for NO3-
B) sp3 for NO2- and sp2 for NO3-
C) sp for NO2- and sp2 for NO3-
D) sp2 for both
23) A molecule with the formula XF3 where the element X has the hybridization sp3. Which of the
following elements could be Y?
A) C
B) P
C) B
D) Si

24) The bonds in the polyatomic ion CO32- are classified as
A) ionic.
B) metallic.
C) nonpolar covalent.
D) polar covalent.
25) The dipole moment of ClF is 0.88 D, and its bond length is 163 pm. What is the percent ionic character
of the Cl—F bond?
A) 0.54%
B) 7.8%
C) 11%
D) 25%
26) The dipole moment of BrF is 1.29 D, and its bond length is 178 pm. What is the percent ionic character
of the Br—F bond?
A) 3.9%
B) 8.5%
C) 15%
D) 33%
27) AgCl is found to have 78.1% ionic character, and its gas phase dipole moment is 11.5 D. What is the
distance between the Ag and Cl atoms in gaseous AgCl?
A) 9.19 × 10-10 pm
B) 14.7 pm
C) 307 pm
D) 903 pm

28) The HI bond has a length of 161 pm and 4.92% ionic character. What is the experimental dipole
moment of HI?
A) 0.380 D
B) 0.772 D
C) 3.80 D
D) 7.72 D
29) Which compound below could have a zero dipole moment?
A) CCl2F2 (tetrahedral)
B) CuCl2F2 (tetrahedral)
C) PtCl2F2 (square planar)
D) SCl2F2 (see-saw)
30) Which of the following molecules has polar bonds but has no net dipole?
A) NH3
B) CCl4
C) CHCl3
D) CH4
31) Which of the following compounds exhibits hydrogen bonding?
A) CH3F
B) CH3OCH3
C) (CH3)3N
D) CH3CH2OH

32) Which substance in each of the following pairs is expected to have the larger dispersion forces?
A) Br2 in set I and n-butane in set II
B) Br2 in set I and isobutane in set II
C) I2 in set I and n-butane in set II
D) I2 in set I and isobutane in set II
33) Which of the following molecules will readily form hydrogen bones with H2O?
A) H2
B) C3H8
C) CH4
D) CH3OH
34) Which molecular orbital resembles a p-orbital?
A) σ
B) σ*
C) π
D) π*

35) If an electron is added to H2 it would go into a
A) σ molecular orbital and strengthen the H—H bond.
B) σ molecular orbital and weaken the H—H bond.
C) σ* molecular orbital and strengthen the H—H bond.
D) σ* molecular orbital and weaken the H—H bond.
36) Which molecular orbital resembles a d-orbital?
A) σ
B) σ*
C) π
D) π*
37) Compare the energies of molecular orbitals of homonuclear diatomic molecules with the energies of
the atomic orbitals with which they correlate.
A) Both bonding and antibonding molecular orbitals lie lower in energy than the atomic orbitals.
B) Bonding orbitals are lower and antibonding orbitals are higher in energy than the atomic orbitals.
C) Bonding orbitals are higher and antibonding orbitals are lower in energy than the atomic orbitals.
D) Both bonding and antibonding molecular orbitals are higher in energy than the atomic orbitals.
38) Which molecular orbitals for homonuclear diatomic molecules are degenerate?
A) π molecular orbitals
B) σ molecular orbitals
C) π molecular orbitals and σ molecular orbitals
D) neither π molecular orbitals nor σ molecular orbitals

39) The MO diagram below is appropriate for B2. Based on this diagram, B2
A) has a bond order of one and is diamagnetic.
B) has a bond order of one and is paramagnetic.
C) has a bond order of two and is diamagnetic.
D) has a bond order of two and is paramagnetic.
40) The following MO diagram is appropriate for Li2 and Be2. Based on this diagram,
A) both are stable and diamagnetic.
B) Li2 is stable and diamagnetic, but Be2 is unstable.
C) Be2 is stable and diamagnetic, but Li2 is unstable.
D) Be2 is stable and paramagnetic, but Li2 is unstable.

41) Given that O2 is paramagnetic and has a bond order of 2, and its highest occupied molecular orbital is
antibonding, what would be the expected bond orders for O22- and O22+?
A) 1 for O22- and 3 for O22+
B) 3/2 for O22- and 5/2 for O22+
C) 5/2 for O22- and 3/2 for O22+
D) 3 for O22- and 1 for O22+
42) The paramagnetism of O2 is explained by
A) coordinate covalent bonding.
B) molecular orbital theory.
C) resonance.
D) valence bond theory.

43) Use the following MO diagram for Be2, Be2+, and Be2-. Based on this diagram,
A) Be2+ is more stable than Be2, and Be2 is more stable than Be2-.
B) Be2- is more stable than Be2, and Be2 is more stable than Be2+.
C) Be2+ and Be2- are both more stable than Be2.
D) Be2 is more stable than either Be2+ or Be2-.
44) Which statement concerning any homonuclear diatomic molecule and its 1- ion must be true?
A) X2 must be more stable than X2-.
B) X2 must be less stable than X2-.
C) X2- must be paramagnetic and X2 must be diamagnetic.
D) X2- must be paramagnetic and X2 may be paramagnetic or diamagnetic.

45) Which is paramagnetic?
A) N22+
B) N22-
C) O22+
D) O22-
46) Based on molecular orbital theory, in order to weaken the N—N bond in N2, electrons should be
A) added.
B) removed.
C) either added or removed.
D) neither added nor removed.
47) In which will the O—O bond be made stronger by removing an electron?
A) only O2
B) only O2-
C) only O22-
D) all of these
48) Molecular orbitals extending over more than two atoms provide an explanation for
A) coordinate covalent bonding.
B) ionic bonding.
C) paramagnetism.
D) resonance.

49) The SO3 molecule can be described as having 3 S—O σ bonds and π molecular orbitals containing
A) 2 electrons.
B) 4 electrons.
C) 6 electrons.
D) 8 electrons.
50) Which is the best description of the bonding in NO3-?
A) 3 N—O σ bonds and no N—O π bonds
B) 3 N—O σ bonds and 3 N—O π bonds
C) no N—O σ bonds and delocalized N—O π molecular orbitals extending over all four atoms
D) 3 N—O σ bonds and delocalized N—O π molecular orbitals extending over all four atoms
51) What is the geometry around the central atom in the following molecular model of NO3-?
A) linear
B) bent
C) trigonal planar
D) trigonal pyramidal

52) What is the geometry around the central atom in the following molecular model of SO2?
A) linear
B) bent
C) trigonal planar
D) trigonal pyramidal
53) What is the geometry around the central atom in the following molecular model of NO2-?
A) linear
B) bent
C) trigonal planar
D) trigonal pyramidal

54) What is the geometry around the central atom in the following molecular model of NH2-?
A) linear
B) bent
C) trigonal pyramidal
D) tetrahedral
55) What is the geometry around the central atom in the following molecular model of SO32-?
A) linear
B) bent
C) trigonal pyramidal
D) tetrahedral
56) What is the geometry around the central atom in the following molecular model of H3O+?
A) linear
B) bent
C) trigonal pyramidal
D) tetrahedral

57) What is the geometry around the central atom in the following molecular model of NH4+?
A) linear
B) bent
C) trigonal pyramidal
D) tetrahedral
58) What is the geometry around the central atom in the following molecular model of SCl4?
A) trigonal bipyramidal
B) seesaw
C) T-shaped
D) linear

59) What is the geometry around the central atom in the following molecular model of BrF3?
A) trigonal bipyramidal
B) seesaw
C) T-shaped
D) linear
60) What is the geometry around the central atom in the following molecular model of XeF2?
A) trigonal bipyramidal
B) seesaw
C) T-shaped
D) linear

61) What is the geometry around the central atom in the following molecular model of I3-?
A) trigonal bipyramidal
B) seesaw
C) T-shaped
D) linear
62) What is the geometry around the central atom in the following molecular model of BrF5?
A) trigonal bipyramidal
B) octahedral
C) square pyramidal
D) square planar

63) What is the geometry around the central atom in the following molecular model of TeF5-?
A) trigonal bipyramidal
B) octahedral
C) square pyramidal
D) square planar
64) What is the geometry around the central atom in the following molecular model of XeF4?
A) trigonal bipyramidal
B) octahedral
C) square pyramidal
D) square planar

65) What is the geometry around the central atom in the following molecular model of ICl4-?
A) trigonal bipyramidal
B) octahedral
C) square pyramidal
D) square planar
66) What are the bond angles in the following molecular model of NO3-?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°
67) What is the bond angle in the following molecular model of SO2?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°

68) What is the bond angle in the following molecular model of NO2-?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°
69) What is the bond angle in the following molecular model of NH2-?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°

70) What are the bond angles in the following molecular model of SO32-?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°
71) What are the bond angles in the following molecular model of H3O+?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°

72) What are the bond angles in the following molecular model of NH4+?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°
73) What are the bond angles in the following molecular model of SCl4?
A) some less than 90° and one less than 180°
B) 90° and 180°
C) some less than 90° one less than 120° but greater than 90°, and one less than 180° but greater than 120°
D) 90°, 120°, and 180°

74) What are the bond angles in the following molecular model of BrF3?
A) two less than 90° and one less than 180° but greater than 90°
B) 90° and 180°
C) two less than 90° and one less than 120° but greater than 90°
D) 90° and 120°
75) What is the bond angle in the following molecular model of XeF2?
A) less than 120°
B) 120°
C) less than 180° but greater than 120°
D) 180°

76) What is the bond angle in the following molecular model of I3-?
A) less than 120°
B) 120°
C) less than 180° but greater than 120°
D) 180°
77) What are the bond angles in the following molecular model of PF6-?
A) some less than 90° and some less than 120° but greater than 90°
B) 90° and 120°
C) some less than 90° and some less than 180° but greater than 120°
D) 90° and 180°

78) What are the bond angles in the following molecular model of BrF5?
A) some less than 90° and some less than 120° but greater than 90°
B) 90° and 120°
C) some less than 90° and some less than 180° but greater than 120°
D) 90° and 180°
79) What are the bond angles in the following molecular model of TeF5-?
A) some less than 90° and some less than 120° but greater than 90°
B) 90° and 120°
C) some less than 90° and some less than 180° but greater than 120°
D) 90° and 180°

80) What are the bond angles in the following molecular model of XeF4?
A) some less than 90° and some less than 120° but greater than 90°
B) 90°
C) some less than 90° and some less than 180° but greater than 90°
D) 90° and 180°
81) What are the bond angles in the following molecular model of ICl4-?
A) some less than 90° and some less than 120° but greater than 90°
B) 90°
C) some less than 90° and some less than 180° but greater than 90°
D) 90° and 180°

82) Shown below is a model of SF6 having an orientation in which one or more atoms are hidden from
view. What is the indicated bond angle?
A) 90°
B) less than 109.5° but greater than 90°
C) less than 120° but greater than 109.5°
D) 120°
83) Identify the set of hybrid orbitals shown below.
A) sp
B) sp2
C) sp3
D) sp4

84) Identify the set of hybrid orbitals shown below.
A) sp
B) sp2
C) sp3
D) sp4
85) Identify the set of hybrid orbitals shown below.
A) sp
B) sp2
C) sp3
D) sp4

86) Which molecule has a central atom that uses the set of hybrid orbitals shown below to form bonds
with the non-central atoms?
A) H2O
B) CO2
C) NO2-
D) ICl2-
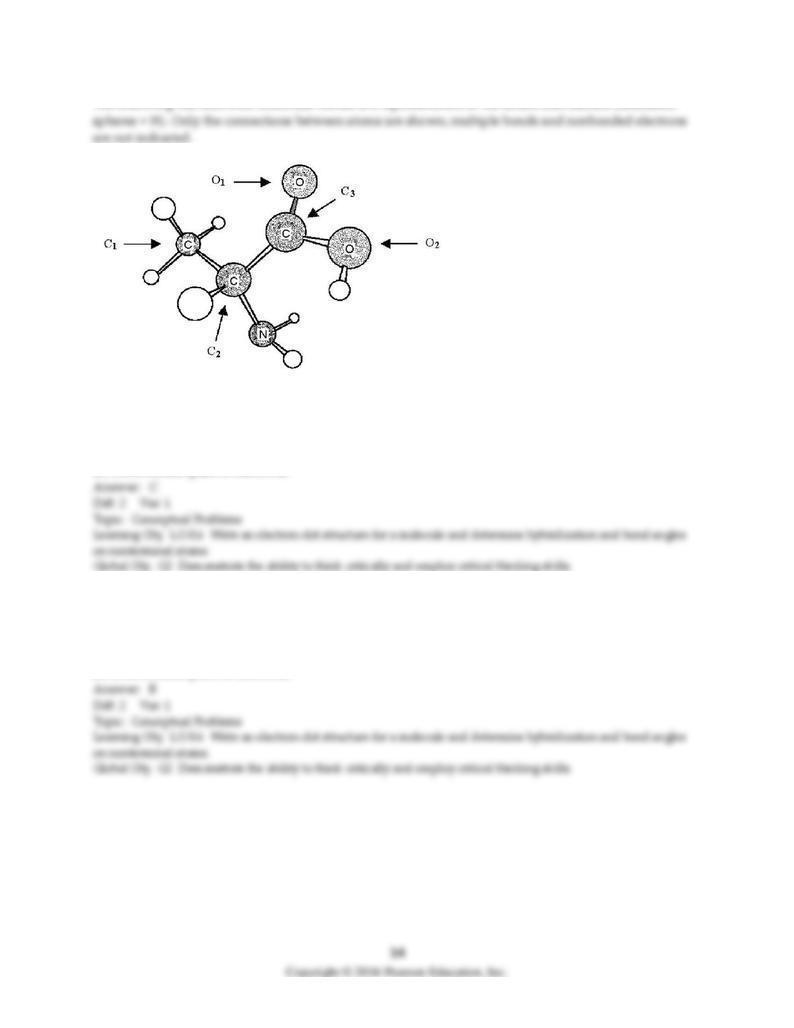
The following ball-and-stick molecular model is a representation of the amino acid alanine (unshaded
87) In order to complete an electron-dot structure of alanine, the nitrogen atom needs
A) 1 additional bond and 1 nonbonded pair of electrons.
B) 1 additional bond and 2 nonbonded pairs of electrons.
C) 1 nonbonded pair of electrons.
D) 2 nonbonded pairs of electrons.
88) In order to complete an electron-dot structure of alanine, the oxygen atom labeled O1 needs
A) 1 additional bond and 1 nonbonded pair of electrons.
B) 1 additional bond and 2 nonbonded pairs of electrons.
C) 1 nonbonded pair of electrons.
D) 2 nonbonded pairs of electrons.

89) In order to complete an electron-dot structure of alanine, the oxygen atom labeled O2 needs
A) 1 additional bond and 1 nonbonded pair of electrons.
B) 1 additional bond and 2 nonbonded pairs of electrons.
C) 1 nonbonded pair of electrons.
D) 2 nonbonded pairs of electrons.
90) What is the hybridization of the carbon atom labeled C1?
A) sp
B) sp2
C) sp3
D) sp4
91) What is the hybridization of the carbon atom labeled C2?
A) sp
B) sp2
C) sp3
D) sp4
92) What is the hybridization of the carbon atom labeled C3?
A) sp
B) sp2
C) sp3
D) sp4

93) What is the hybridization of the oxygen atom labeled O1?
A) sp
B) sp2
C) sp3
D) sp4
94) What is the hybridization of the oxygen atom labeled O2?
A) sp
B) sp2
C) sp3
D) sp4
95) What is the hybridization of the nitrogen atom?
A) sp
B) sp2
C) sp3
D) sp4
96) What is geometry around the carbon atom labeled C1?
A) bent
B) tetrahedral
C) trigonal planar
D) trigonal pyramidal

97) What is geometry around the carbon atom labeled C2?
A) bent
B) tetrahedral
C) trigonal planar
D) trigonal pyramidal
98) What is geometry around the carbon atom labeled C3?
A) bent
B) tetrahedral
C) trigonal planar
D) trigonal pyramidal
99) What is geometry around the oxygen atom labeled O2?
A) bent
B) tetrahedral
C) trigonal planar
D) trigonal pyramidal
100) What is geometry around the nitrogen atom?
A) bent
B) tetrahedral
C) trigonal planar
D) trigonal pyramidal

101) A molecular model of NO3- is shown below. Based on the best Lewis electron-dot structure for NO3-
and formal charge considerations, what is the predicted N—O bond order for each N—O bond?
A) 1
B) 1.33
C) 1.5
D) 2
102) A molecular model of BBr3 is shown below. Based on the best Lewis electron-dot structure for BBr3
and formal charge considerations, what is the predicted B—Br bond order for each B—Br bond?
A) 1
B) 1.33
C) 1.5
D) 2

103) A molecular model of BeF2 is shown below. Based on the best Lewis electron-dot structure for BeF2
and formal charge considerations, what is the predicted Be—F bond order for each Be—F bond?
A) 1
B) 1.33
C) 1.5
D) 2
104) A molecular model of SO42- is shown below. Based on the best Lewis electron-dot structure for
SO42- and formal charge considerations, what is the predicted S—O bond order for each S—O bond?
A) 1
B) 1.33
C) 1.5
D) 2

105) Electrostatic potential maps use color to portray the calculated electron distribution in a molecule.
Atoms that are electron poor and carry a δ+ charge are shown in blue. Atoms that are electron rich and
carry a δ- charge are shown in red. Atoms with little or no charge are shown in green. The electrostatic
potential map of H2O below should show
A) H blue and O red.
B) H blue and O green.
C) H green and O blue.
D) H red and O blue.
106) Electrostatic potential maps use color to portray the calculated electron distribution in a molecule.
Atoms that are electron poor and carry a δ+ charge are shown in blue. Atoms that are electron rich and
carry a δ- charge are shown in red. Atoms with little or no charge are shown in green. The electrostatic
potential map of CH3Cl below should show
A) C blue and Cl red.
B) C blue and Cl green.
C) C green and Cl blue.
D) C red and Cl blue.

107) Electrostatic potential maps use color to portray the calculated electron distribution in a molecule.
Atoms that are electron poor and carry a δ+ charge are shown in blue. Atoms that are electron rich and
carry a δ- charge are shown in red. Atoms with little or no charge are shown in green. The electrostatic
potential map of CH3Li below should show
A) C blue and Li red.
B) C blue and Li green.
C) C green and Li blue.
D) C red and Li blue.
108) In the drawing of acetaldehyde, CH3CHO, the largest partial positive charge (δ+) occurs on
A) atom (a).
B) atom (b).
C) atom (c).
D) atom (d).

109) In the drawing of acetaldehyde, CH3CHO, the largest partial negative charge (δ-) occurs on
A) atom (a).
B) atom (b).
C) atom (c).
D) atom (d).
110) In the drawing of acetic acid, CH3CO2H, a partial positive charge (δ+) occurs on
A) only atom (a).
B) only atom (b).
C) atoms (a) and (c).
D) atoms (b) and (d).
111) In the drawing of acetic acid, CH3CO2H, a partial negative charge (δ-) occurs on
A) only atom (a).
B) only atom (b).
C) atoms (a) and (c).
D) atoms (b) and (d).
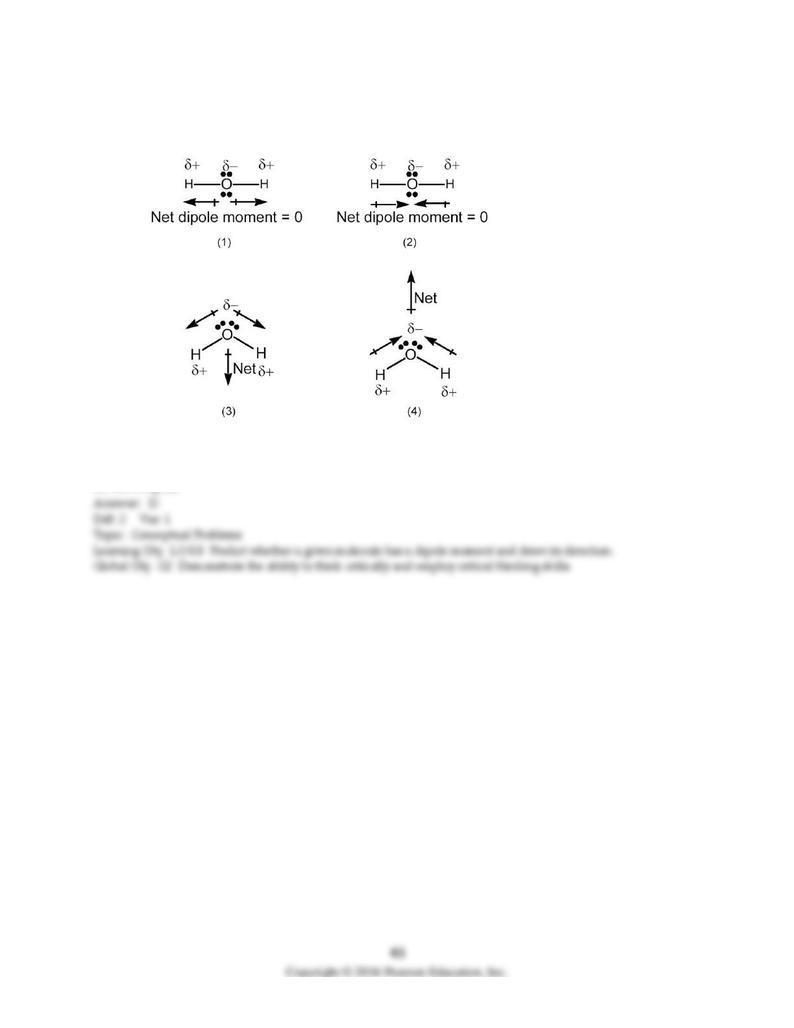
112) Which drawing best accounts for the polarity of water, H2O, and the bond polarities that make a
major contribution to the overall molecular polarity?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)
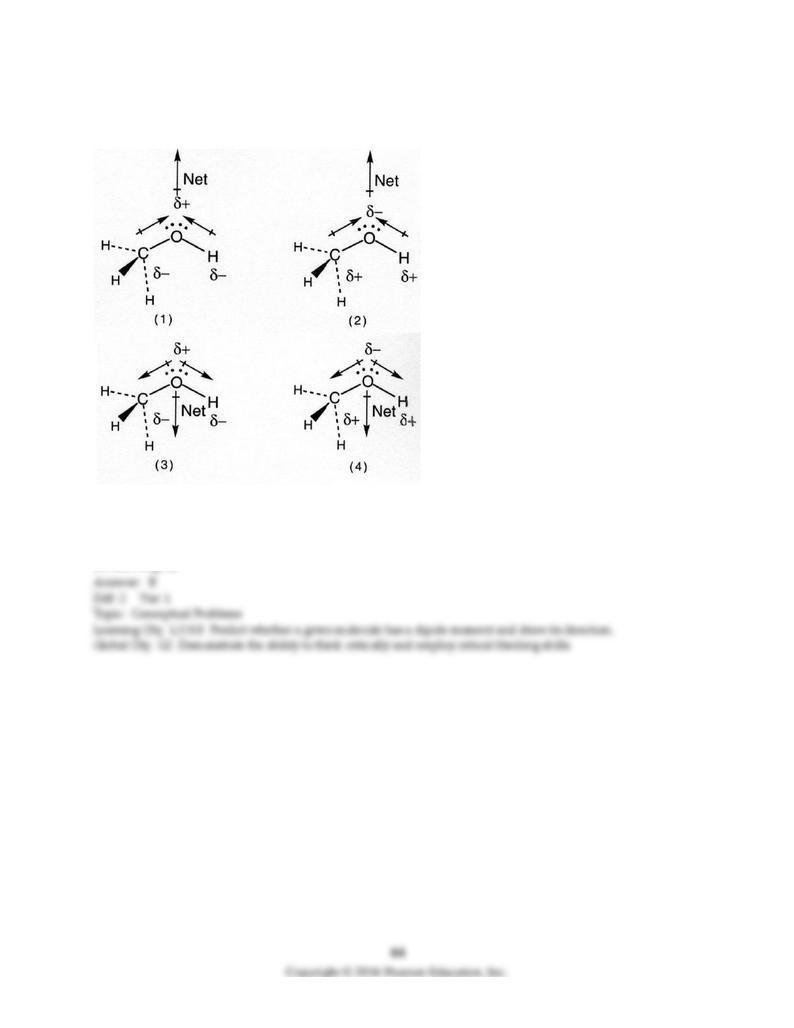
113) Which drawing best accounts for the polarity of methanol, CH3OH, and the bond polarities that
make a major contribution to the overall molecular polarity?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)
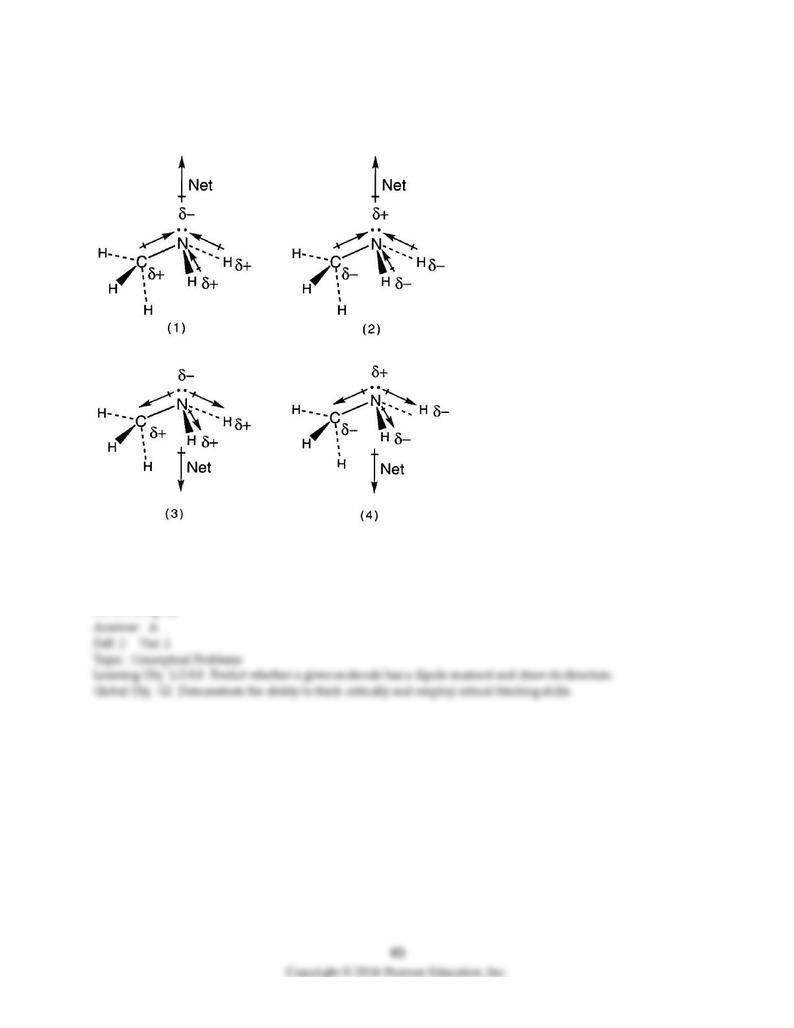
114) Which drawing best shows the molecular polarity of methylamine, CH3NH2, and the bond
polarities that make a major contribution to the overall molecular polarity?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)

115) Which drawing best shows the direction of the dipole moment in H2C=CCl2?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)
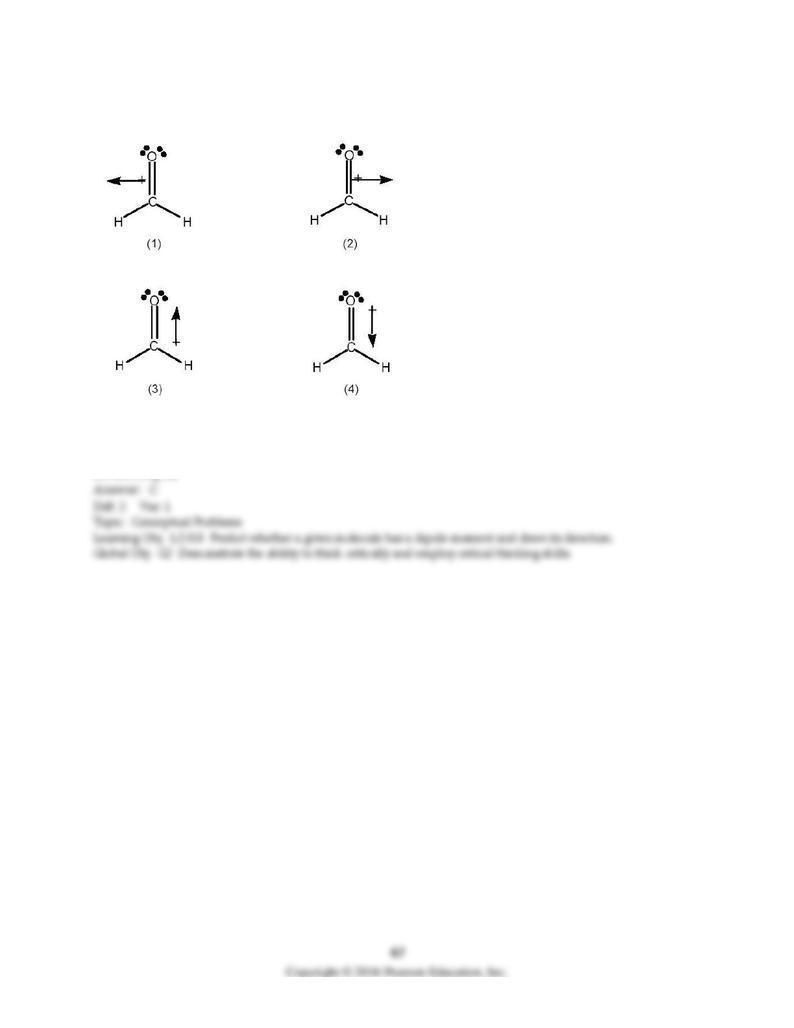
116) Which best indicates the direction of the dipole moment in formaldehyde, H2C=O?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)
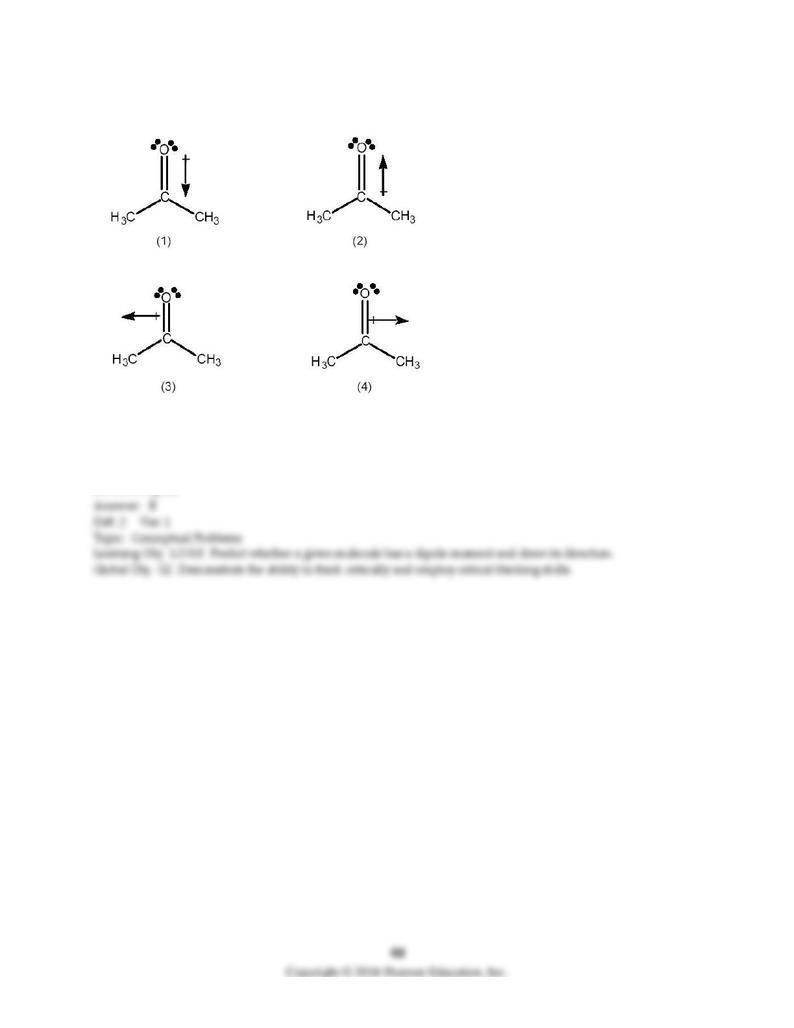
117) Which best indicates the direction of the dipole moment in acetone, (CH3)2C=O?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)
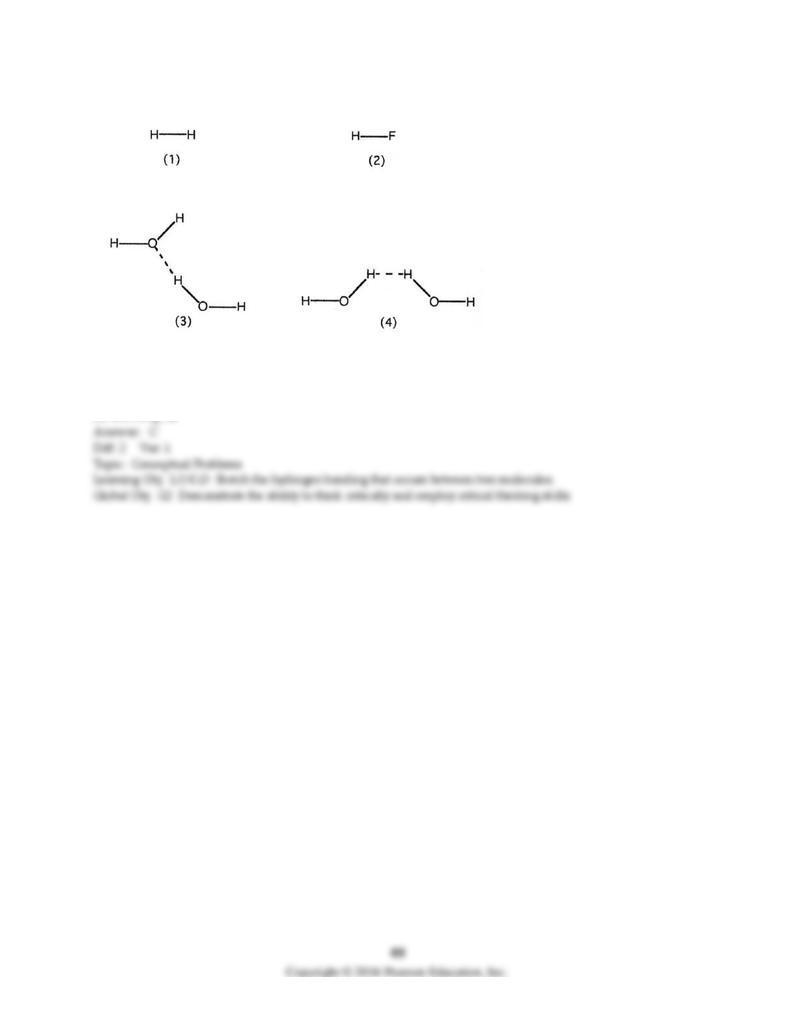
118) Which drawing best represents hydrogen bonding?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)

119) Which drawing below best represents hydrogen bonding methanol, CH3OH?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)

120) Which drawing best represents hydrogen bonding in methylamine, CH3NH2?
A) drawing (1)
B) drawing (2)
C) drawing (3)
D) drawing (4)
121) Which drawing represents a σ bonding molecular orbital for a homonuclear diatomic molecule?
A)
B)
C)
D)

122) Which drawing represents a σ* antibonding molecular orbital for a homonuclear diatomic molecule?
A)
B)
C)
D)
123) Which drawing represents a π* antibonding molecular orbital for a homonuclear diatomic molecule?
A)
B)
C)
D)

124) Which drawing represents a π bonding molecular orbital for a homonuclear diatomic molecule?
A)
B)
C)
D)
125) Which drawing represents the molecular orbital containing the highest energy electrons in the H2
molecule in its ground state?
A)
B)
C)
D)

126) Which drawing represents the molecular orbital containing the highest energy electrons in the O2
molecule in its ground state?
A)
B)
C)
D)
127) Which drawing represents the lowest energy unoccupied molecular orbital in the N2 molecule in its
ground state?
A)
B)
C)
D)

128) Which drawing represents the molecular orbital containing the highest energy electrons in the F2
molecule in its ground state?
A)
B)
C)
D)
129) Which drawing represents the molecular orbital containing the highest energy electrons in the O22-
molecular ion in its the ground state?
A)
B)
C)
D)

8.2 Algorithmic Questions
1) What geometric arrangement of charge clouds is expected for an atom that has five charge clouds?
A) tetrahedral
B) square planar
C) trigonal bipyramidal
D) octahedral
2) What geometric arrangement does the molecule have for a central atom that has five charge clouds
with two lone pairs?
A) tetrahedral
B) octahedral
C) T-shaped
D) trigonal bipyramidal
3) What is the molecular geometry of ICl4-?
A) seesaw
B) square planar
C) square pyramidal
D) tetrahedral
4) What is the molecular geometry of SbCl3?
A) T-shaped
B) tetrahedral
C) trigonal planar
D) trigonal pyramidal

5) What is the O—S—O bond angle in SO3?
A) less than 109.5°
B) 109.5°
C) 120°
D) greater than 120°
6) What is the molecular geometry of SeF5-?
A) octahedral
B) seesaw
C) square pyramidal
D) trigonal bipyramidal
7) Which of the following best describes ICl2-? It has a molecular geometry that is
A) linear with no lone pairs on the I atom.
B) linear with lone pairs on the I atom.
C) nonlinear with no lone pairs on the I atom.
D) nonlinear with lone pairs on the I atom.
8) What is the molecular geometry of TeBr4?
A) seesaw
B) square planar
C) square pyramidal
D) tetrahedral

9) What are the F—Se—F bond angles in SeF6?
A) 60°
B) 90°
C) 109.5°
D) 120°
10) The VSEPR model predicts the O—S—O bond angle in SO2 to be
A) 90°.
B) 109.5°.
C) less than 120° but greater than 109.5°.
D) 120°.
11) Arrange in order from the smallest to the largest bond angle: CCl3+, NF3, NH4+, XeBr4.
A) CCl3+, NF3, NH4+, XeBr4
B) NF3, NH4+, XeBr4, CCl3+
C) XeBr4, NH4+, NF3, CCl3+
D) XeBr4, NF3, NH4+, CCl3+
12) The VSEPR model predicts the H—B—H bond angle in BH3 to be
A) 90°.
B) 109.5°.
C) less than 120° but greater than 109.5°.
D) 120°.

13) What is the electron geometry and molecular shape of CH2O?
A) Tetrahedral, tetrahedral
B) Trigonal planar, trigonal planar
C) Trigonal planar, bent
D) Tetrahedral, trigonal planar
14) What electron geometry would you expect for atoms that have 6 charge clouds?
A) Tetrahedral
B) Octahedral
C) Trigonal planar
D) Trigonal bipyramid
15) A single sp3 hybrid orbital has
A) one lobe.
B) two lobes of equal size.
C) two lobes of unequal size.
D) three lobes of equal size.
16) What orbital hybridization is expected for the central atom in a molecule with a trigonal planar
geometry?
A) sp
B) sp2
C) sp3
D) sp4

17) The orbital hybridization on the carbon atom in S CN - is
A) sp.
B) sp2.
C) sp3.
D) sp3d2.
18) Which covalent bond is the most polar?
A) Cl—F
B) B—F
C) O—F
D) F—F
19) Which of the following should have the largest dipole moment?
A) H2(g)
B) CO2(g)
C) KCl(g)
D) CH3F(g)
20) Which of the following molecules does not have a dipole moment?
A) C2H4
B) NH3
C) CH3NH2
D) HCl

21) Which covalent bond is the most polar?
A) H—I
B) H—F
C) H—Cl
D) H—Br
22) Which has a dipole moment?
A) CF4
B) SiO32-
C) SO2
D) SO42-
23) Which of the following molecules does not have a dipole moment?
A) CBr2=CBr2
B) NCl3
C) CH3NH2
D) HCl
24) Which of the following should have the largest dipole moment?
A) F2(g)
B) SO2(g)
C) RbBr(g)
D) CH2I2(g)

25) Which compound, shown with its dipole moment, is expected to exhibit the smallest percent ionic
character?
A) CCl4
B) HBr
C) KCl
D) CH2Cl2
26) Which has the smallest dipole-dipole forces?
A) CH3Cl
B) KCN
C) I2
D) H2S
27) Which has the smallest dipole-dipole forces?
A) CH3F
B) HCl
C) N2
D) CO
28) Which of the following exhibits ion-dipole forces?
A) KBr(s)
B) NaF(aq)
C) Ag(s)
D) Cl2(g)

29) Which is expected to have the largest dispersion forces?
A) C2H4
B) C18H36
C) NBr3
D) Ba
30) Which is expected to have the largest dispersion forces?
A) C2H4
B) C8H16
C) Cl2
D) N2H4
31) Which of the following compounds exhibits hydrogen bonding?
A) CH3I
B) HI
C) CH3OCH3
D) CH3NH2
32) Which of the following compounds has the highest boiling point?
A) H2O
B) H2S
C) H2Se
D) H2Te

33) Which of the following compounds exhibits only dispersion and dipole-dipole intermolecular
interactions?
A) Na
B) HCl
C) BH3
D) CH3NHC4H9
34) In C3H7NHC3H7, which intermolecular forces are present?
A) Dispersion, hydrogen bonding and dipole-dipole forces are present.
B) Only dipole-dipole and ion-dipole forces are present.
C) Only dispersion and dipole-dipole forces are present.
D) Only hydrogen bonding forces are present.
35) In liquid pentanol, CH3CH2CH2CH2 , which intermolecular forces are present?
A) Dispersion, hydrogen bonding and dipole-dipole forces are present.
B) Only dipole-dipole and ion-dipole forces are present.
C) Only dispersion and dipole-dipole forces are present.
D) Only hydrogen bonding forces are present.
36) Which of the following compounds exhibits only dispersion and dipole-dipole intermolecular
interactions?
A) N2
B) HBr
C) CO2
D) H2O

37) The normal boiling point for H Br is higher than the normal boiling point for H Cl. This can be
explained by
A) larger dipole-dipole forces for H Br.
B) larger dispersion forces for H Br.
C) larger hydrogen-bond forces for H Br.
D) larger dipole-dipole forces, larger dispersion forces, and larger hydrogen-bond forces for H Br.
38) What is the geometry around the central atom in the following molecular model of BF3?
A) linear
B) bent
C) trigonal planar
D) trigonal pyramidal
39) What is the geometry around the central atom in the following molecular model of H2Se?
A) linear
B) bent
C) trigonal pyramidal
D) tetrahedral

40) What is the geometry around the central atom in the following molecular model of PH3?
A) linear
B) bent
C) trigonal pyramidal
D) tetrahedral
41) What is the geometry around the central atom in the following molecular model of CCl4?
A) linear
B) bent
C) trigonal pyramidal
D) tetrahedral

42) What is the geometry around the central atom in the following molecular model of PF5?
A) trigonal bipyramidal
B) seesaw
C) T-shaped
D) linear
43) What is the geometry around the central atom in the following molecular model of SI6?
A) trigonal bipyramidal
B) octahedral
C) square pyramidal
D) square planar

44) What is the geometry around the central atom in the following molecular model of PCl6-?
A) trigonal bipyramidal
B) octahedral
C) square pyramidal
D) square planar
45) What are the bond angles in the following molecular model of BBr3?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°

46) What is the bond angle in the following molecular model of H2Te?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°
47) What are the bond angles in the following molecular model of PH3?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°

48) What are the bond angles in the following molecular model of SiCl4?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°
49) What are the bond angles in the following molecular model of PCl5?
A) some less than 90° and one less than 180°
B) 90° and 180°
C) some less than 90° and some less than 120° but greater than 90°, and one less than 180° but greater
than 120°
D) 90°, 120°, and 180°

50) What are the bond angles in the following molecular model of SI6?
A) some less than 90° and some less than 120° but greater than 90°
B) 90° and 120°
C) some less than 90° and some less than 180° but greater than 120°
D) 90° and 180°
51) Shown below is a model of CBr4 having an orientation in which one or more atoms are hidden from
view. What is the indicated bond angle?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°

52) Shown below is a model of SiH4 having an orientation in which one or more atoms are hidden from
view. What is the indicated bond angle?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°
53) Shown below is a model of PCl5 having an orientation in which one or more atoms are hidden from
view. What is the indicated bond angle?
A) less than 109.5°
B) 109.5°
C) less than 120° but greater than 109.5°
D) 120°

54) Which molecule has a central atom that uses the set of hybrid orbitals shown below to form bonds
with the non-central atoms?
A) H2S
B) CS2
C) NO2-
D) IF2-
55) Which molecule has a central atom that uses the set of hybrid orbitals shown below to form bonds
with the non-central atoms?
A) H2Se
B) CS2
C) NO2-
D) ICl2-

8.3 Short Answer Questions
1) The molecular geometry of OCCl2 is ________.
2) Of XeF2 and XeF4, the one with the smaller bond angles is ________.
3) Of BrF3 and PF3, the one with the smaller bond angles is ________.
4) Of NH4+ and NH4- the one with the smaller bond angles is ________.
5) The hybrid orbital used by nitrogen to overlap with the 1s orbital of hydrogen in CH3NH2 is ________.
6) The carbon-carbon bond in C2H2 contains ________ σ and ________ π bonds.

7) The hybrid orbital used by carbon to overlap with hydrogen in C2H2 is ________.
8) The bonds in the polyatomic ion NO3- are classified as ________.
9) In the molecule BF3 there is a δ+ charge on the ________ atom and a δ- charge on the ________ atom.
10) LiH has an experimental dipole moment, μ = 6.00 D. If LiH were 100% ionic, the distance between
positive and negative charges would be 161 pm. What is the percent ionic character in the LiH bond?
11) The Br—Cl bond has 5.05% ionic character and a dipole moment of 0.518 D. What is the distance
between atoms in BrCl?
12) The HBr bond has a length of 141 pm and 12.1% ionic character. What is the dipole moment of HBr?

13) Pt(NH3)2Cl2 is square planar. The isomer of Pt(NH3)2Cl2 that has a non-zero dipole moment has a
bond angle of ________ degrees.
14) The polarity of CCl4 is ________.
15) The intermolecular forces formed when KI is dissolved in water are ________ forces.
16) The intermolecular forces responsible for CH3CH2OH being at liquid at 20°C are ________ bonds.
17) Helium can be liquefied when He atoms are attracted to one another by intermolecular ________
forces.
18) According to molecular orbital theory, is the highest energy orbital that contains an electron
antibonding or bonding in O22-?

19) When an electron is added to the lowest unoccupied molecular orbital of N2, the electron is added to
a(n) ________ (antibonding, bonding) molecular orbital and the N—N bond order will ________
(decrease, increase).
20) Predict whether removing two electrons from F2 will create an ion that is diamagnetic or
paramagnetic and have an F—F that is stronger or weaker than the bond in F2.
21) Are the π-bonds in CH3CO2- delocalized or localized?